Monoterpenoids and Their Synthesized Brominate Derivatives as Eco-Friendly Measures to Control Some Plant Pathogenic Fungi and Bacteria
| Received 19 Apr, 2023 |
Accepted 20 Jul, 2023 |
Published 30 Sep, 2023 |
Background and Objective: Phyto-pathogenic fungi or bacteria are one of the most important causes that decrease food and cash crops. Chemical pesticides cause damage to the environment. Thus, globally, there are attempts to provide eco-friendly products. Therefore, this work aimed to find alternative natural products for managing serious diseases (fungi and bacteria). Materials and Methods: Fungicidal and bactericidal of four monoterpenoids, as well as their synthesized brominated (Br) derivatives were investigated against six fungi and four bacteria in vitro. The synthesized brominated derivatives were confirmed by a mass spectrometer and GC/MS. Results: The soil and air-borne fungi were very sensitive to chlorothymol followed by thymol and carvacrol, respectively. However, the synthesized brominate derivatives such as bromocarvacrol were the most effective treatment against all the tested fungal strains, while, bromoegunol was a less effective treatment. Concerning the antibacterial activity, the tested monoterpenoids exhibited variable degrees of antibacterial against all of the tested bacterial strains and the conversion of monoterpenoids into bromo-derivatives enhanced the bactericidal activity. Conclusion: The chlorothymol (monoterpene) recorded the highest fungicidal activity against soil and air-born fungi. For bacterial activity, results exhibited that chlorothymol was the most effective monoterpene against bacteria. All the bromo-derivatives showed high activity against the most of tested fungi and bacteria.
| Copyright © 2023 Ahmed et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Biopesticides are considered a unique solution to the problem of agrochemicals in crop protection. It reaches the target pest leaving the remaining 99.9% to enter the environment to cause hazards to non-target organisms including humans1. In the last two decades, scientists all over the world tried to minimize the use of synthetic pesticides for the management of plant pathogens, insects, acari and weeds to avoid environmental pollution hazards. Besides, the targeted pathogen, pesticides may also kill various beneficial organisms. The increasing incidence of resistance among pathogens towards synthetic chemicals is also a cause of serious concern2. Major food and cash crops are attacked by certain pathogens especially fungal diseases which cause approximately 20% reductions in the yield3,4.
The discovery of antimicrobial compounds from plant sources is a safe route to a new generation characterized by eco-friendly, low toxicity, selectivity, highest efficacy and biodegradability5-7.
Search extensively for new biologically active terpenoids as a potential source for agrochemicals. Monoterpenes together with sesquiterpenes and diterpenes form the majority of essential oil. Monoterpenes are unsaturated hydrocarbons and some are oxygenated derivatives such as alcohol, ketones, carboxylic acid and phenol8,9. Chemical modification of natural monoterpenoids to various derivatives has been reported to result in modification of biological activity10-14. Therefore, this work was one of several attempts that have been conducted to find alternative natural products which are safe, biodegradable and eco-friendly for managing serious pests (fungi and bacteria) which attack crop plants.
MATERIALS AND METHODS
In this investigation, the evaluated monoterpenoids and/or their synthesized brominated derivatives were implemented under laboratory conditions in 2021 at the Fungicide Bioassay Laboratory, Department of Pesticide Chemistry and Technology, Faculty of Agriculture, University of Alexandria, Alexandria, Egypt.
Tested monoterpenoids: The tested monoterpenoids were thymol, chlorothymol, eugenol and carvacrol. All monoterpenoids were purchased from Aldrich Chemicals Ltd., UK.
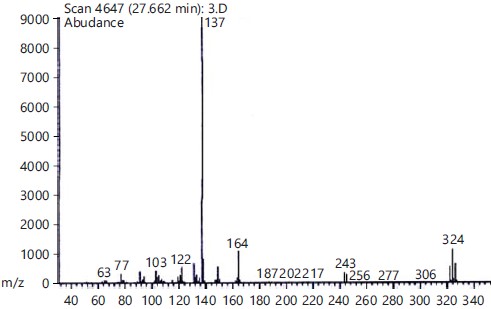
General procedure for bromination of tested monoterpenoids: The desired monoterpenoids (0.01 mol) thymol, eugenol, chlorothymol and carvacrol were dissolved in carbon disulfide (25 mL) in a round bottom flask fitted with a reflux condenser and pressure equivalent-dropping funnel. Bromine (0.01 mol) was dissolved in carbon disulfide (16 mL) and then added dropwise to a cooled flask (below 5°C) after the reaction mixture was stirred at room temperature for 1hr to complete (Scheme 1) and then monitored by thin layer chromatography (TLC with aluminum plate of 25, silica gel coated with fluorescent indicator F254, TLC size is 20×20 cm, MERCK, Germany). The reaction was quenched with water and extracted three times with methylene chloride (30 mL each). The organic layer was washed three times with water (100 mL) and dried over anhydrous sodium sulfate (50 g). The organic solvent was removed using a rotary evaporator under a high vacuum (brand name: Kryqcn, Model: RE501, rotating speed: 120 rpm, vacuum power: 0.098 mpa, China). The product was identified by a mass spectrometer (HP model, MS-5988) and GC/MS (Shimadzu QP2010) and Melting points were measured with capillary melting point apparatus model 1002-USA, UV spectra were recorded on a UV Spectrophotometer model UV-1601 SHIMADZU shown in Table 1 and 2.

|
| Table 1: | Physical properties of the monoterpenoidal derivatives | |||
| Derivative | Purity (%) |
M.P (°C ) |
| Bromothymol | 87 |
48 |
| Bromochlorothymol | 85 |
Oil |
| Bromocarvacrol | 97 |
46 |
| Bromoeugenol | 95 |
Oil |
| M.P: Melting point | ||
| Table 2: | Identification of the synthesized monoterpenoidal brominate derivatives | |||
| Compound | Empirical formula |
Estimated M.W. |
Found M.W. |
|
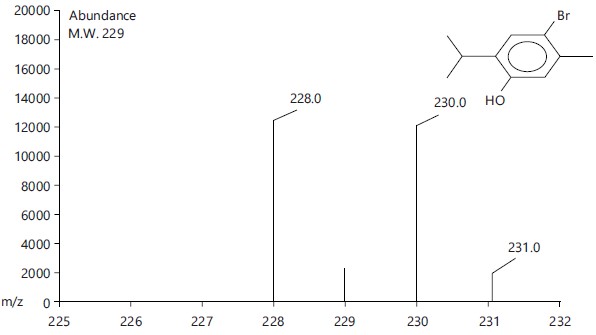
| Bromothymol |  |
C10H13OBr |
229.11 |
229 |
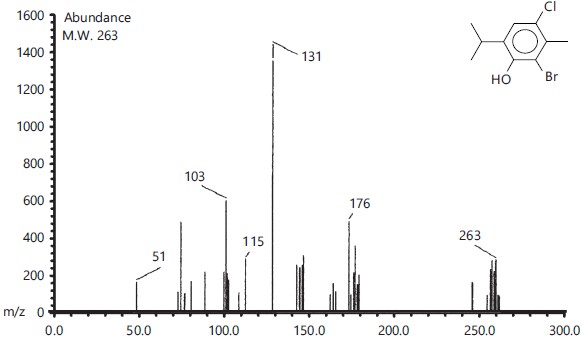
| Bromocholrothymol |  |
C10H12OclBr |
263.56 |
263 |
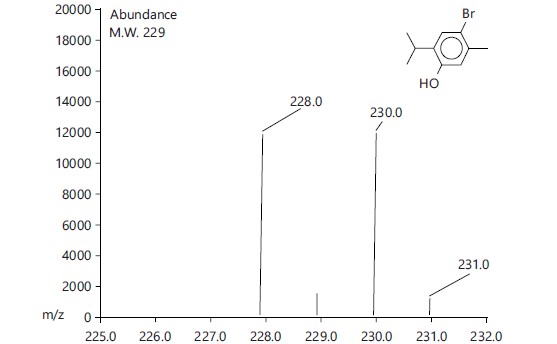
| Bromocarvacrol |  |
C10H13Obr |
229.11 |
229 |
| Bromoeugenol |  |
C10H11O2Br |
243.1 |
243 |
| M.W.: Molecular weight | ||||
Antifungal assay: Six economic plant pathogenic fungi were chosen for this study. Fungi were obtained from the Fungicide Bioassay Laboratory, Department of Pesticide Chemistry, Faculty of Agriculture, Alexandria University. However, the studied fungi were as follows:
| • | Soil borne fungi: Fusarium oxyporum, Rhizoctonia solani and Pythium debrinum | |
| • | Airborne fungi: Alternaria alternata, Helminthosporium sp. and Botrytis feba |
The antifungal activity of tested compounds was investigated by using the radial growth technique method15. Appropriate volumes of the stock solutions of the tested compounds either in dimethyl sulfoxide (DMSO) for solvent extracts or in distilled water for water extracts were added to molten nutrient agar (potato dextrose agar medium, PDA) to achieve the desired concentration immediately before pouring into the Petri dishes (9.0 cm in diameter) at 40-45°C.
Each concentration was tested in triplicate. Parallel controls were maintained with DMSO mixed with PDA. The discs of mycelial felt (0.5 cm diameter) of the plant pathogenic fungi, taken from 8-day-old cultures on PDA plates, were transferred aseptically to the center of Petri dishes. The treatments were incubated at 25°C in the dark. Colony growth diameter was measured after the fungal growth in the control treatments which completely covered the Petri dishes. Moreover, the percentage of mycelial growth inhibition was calculated according to the following formula16:
where, DC and DT are the average diameters of the fungal colony of control and treatment, respectively.
Bactericidal activity assay: Four phytopathogenic bacteria, Agrobacterium tumefaciens (Crown gall bacteria), Erwinia carotovra subsp. Carotovra (soft rot bacteria), Erwinia amylovora (fire blight bacteria) and Pseudomonas solanacearum were provided by the Department of Plant Pathology, Faculty of Agriculture, Alexandria University, Egypt. The bacterial strains were cultured in a glycerol agar medium. Streptomycin sulfate as a standard bactericide was supplied by El-Nile Company for Chemical Industry and Drugs, Egypt.
Determination of minimum inhibitory concentrations (MICs): Tested compounds were dissolved in dimethyl sulfoxide (DMSO). Appropriate volumes of the stock solution were added to nutrient agar and poured into Petri dishes (diameter 9 mm). After solidification, a bacterial culture has grown in a nutrient broth for 18 hrs (approximately 108 CFU mL–1 was planted on four lines per each plate with three replicates from four bacteria species Agrobacterium tumefaciens, Erwinia carotovra, Erwinia amylovora and Pseudomonas salnacearum on the surface of the agar. The inoculum line was allowed to dry before inverting the plates for incubation at 27°C for 24 hrs. The control was nutrient agar with a maximum volume of dimethylsulfoxide added to the treatment. The antibiotic, streptomycin was used for comparison, The MIC was determined as the lowest concentration of the tested compound showing no visible bacterial growth in the agar plates. As recommended by the European Society of Clinical Microbiology and Infection Disease17.
Statistical analysis: The effective concentrations of evaluated compounds that inhibit the fungi mycelia or bacteria growth by 50% (EC50), were conducted by a linear regression method with probit analysis18.
RESULTS AND DISCUSSION
Identification of mono terpenoids derivatives: The reaction of thymol, carvacrol, chlorothymol and eugenol with carbon disulfide and bromine gave bromothymol and bromocarvacrol as crystals, while bromocholorothymol and bromoeugenol as oily compounds. The percentage of the product yield obtained from the reaction was 80, 83, 92 and 90% for Bromothymol, Bromochlorothymol, Bromocarvacrol and Bromoeugenol, respectively. Physical properties and identification of the synthesized monoterpenoidal brominate derivatives showed in Table (1, 2) and Fig. (1-4).
|
|
|
Fungicidal activity of some monoterpenoids towards soil and air-borne fungi: The fungicidal activity of four monoterpenoids namely, carvacrol, chlorothymol, eugenol and thymol were tested against soil-born fungi (fusarium oxysprum, Rhizoctonia solani and Pythium debrinum) and air-born fungi (Botrytis feba, Helimanthosporum sp. and Alteranria alternate) as shown in Table 3. The fungus F. oxysproum was very sensitive to chlorothymol, thymol, carvacrol and eugenol with EC50 values estimated by 13.40, 40.57, 140.34 and 271.73 mg L–1, respectively, while Rhizoctonia solani was sensitive to chlorothymol (EC50 = 21.00 mg L–1), carvacrol (EC50 = 21.88 mg L–1), thymol (EC50 = 30.90 mg L–1) and eugenol (EC50 = 140.33). The soil-born fungus, P. debrinum recorded EC50 values estimatedat 28.39, 43.00, 100.88 and 300 mg L–1 with chlorothymol, thymol, carvacrol and eugenol, respectively. However, the standard fungicide (mancozeb) recorded EC50 values of 140.34, 154.59 and 91.70 mg L–1 with F. oxysproum, R. solani and P. debrinum, respectively.
|
| Table 3: | Fungicidal activity of tested monoterpenoids against phytopathogenic fungi | |||
95% confidence limits |
||||
| Fungus | EC50/ppm |
Lower |
Upper |
Slope±SE |
| Chlorothymol | ||||
| Fusarium oxysporum | 13.4 |
11.2 |
18.16 |
2.01±0.3 |
| Rhizoctonia solani | 21 |
15.5 |
31.22 |
2.00±0.19 |
| Pythium debrinum | 28.39 |
23.8 |
32.47 |
3.10±0.35 |
| Alteranria altranata | 7.58 |
5.31 |
8.44 |
1.10±0.18 |
| Helimanthosporum sp. | 4.49 |
0.21 |
7.09 |
2.60±0.35 |
| Botrytis febae | 3.8 |
0.84 |
5.12 |
1.40±0.16 |
| Thymol | ||||
| Fusarium oxysporum | 40.57 |
15.53 |
86.12 |
2.00±0.18 |
| Rhizoctonia solani | 30.9 |
27.14 |
34.84 |
2.60±0.24 |
| Pythium debrinum | 43 |
27.7 |
73.73 |
1.72±0.11 |
| Alteranria altranata | 27.82 |
15.61 |
42.7 |
2.80±0.26 |
| Helimanthosporum sp. | 17.38 |
9.66 |
100.14 |
2.30±0.29 |
| Botrytis febae | 22.97 |
8.85 |
40.88 |
2.40±0.3 |
| Carvacrol | ||||
| Fusarium oxysporum | 140.34 |
122.59 |
160.04 |
2.00±0.24 |
| Rhizoctonia solani | 21.85 |
16.59 |
27.44 |
1.60±0.19 |
| Pythium debrinum | 100.88 |
91.16 |
112 |
3.50±0.3 |
| Alteranria altranata | 18.8 |
12.4 |
25.8 |
0.78±0.13 |
| Helimanthosporum sp. | 53.96 |
22.92 |
93.62 |
1.90±0.2 |
| Botrytis febae | 23.99 |
14.98 |
30.97 |
1.60±32 |
| Eugenol | ||||
| Fusarium oxysporum | 271.73 |
217.01 |
403.75 |
2.68±0.4 |
| Rhizoctonia solani | 140.33 |
121.92 |
167.21 |
2.59±0.33 |
| Pythium debrinum | >300 |
- |
- |
- |
| Alteranria altranata | >300 |
- |
- |
0.80±0.20 |
| Helimanthosporum sp. | 142.9 |
109.81 |
224.94 |
1.27±0.3 |
| Botrytis febae | 138.7 |
123.7 |
158.55 |
3.30±0.37 |
| Mancozeb | ||||
| Fusarium oxysporum | 140.34 |
122.59 |
160.04 |
2.00±0.24 |
| Rhizoctonia solani | 154.59 |
124.82 |
192.62 |
1.18±0.11 |
| Pythium debrinum | 91.7 |
61.93 |
294.8 |
0.90±0.28 |
| Alteranria altranata | 115.61 |
77.65 |
170.97 |
0.62±0.09 |
| Helimanthosporum sp. | 47.44 |
30.83 |
56.37 |
0.77±0.09 |
| Botrytis febae | 236.37 |
127.53 |
612.23 |
1.35±0.1 |
| SE: Standard error | ||||
On the other hand, results showed that the air-born fungus A. alternate was very sensitive to chlorothymol (EC50= 7.58 mg L–1) followed by carvacrol (EC50 = 18.80 mg L–1), thymol (EC50 = 27.82 mg L–1) and eugenol (EC50 = 300 mg L–1). Helimanthosporum sp., was sensitive towards chlorothymol, thymol, carvacrol and eugenol and recorded EC50 values estimated by 4.49, 17.38, 53.96 and 142.90 mg L–1, respectively. The air-born fungus B. febae was respectively very sensitive to chlorothymol, thymol, carvacrol and eugenol, with EC50 values of 3.80, 22.97, 23.99 and 138.70 mg L–1. The estimated EC50 values of mancozeb against A. alternate, Helimanthosporum sp. and B. febae were 115.61, 47.44 and 236.37 mg L–1, respectively.
The obtained results indicated that carvacrol, thymol, cholorothymol and eugenol exhibited the highest antifungal activity against phytopathogenic fungi19-22. The activity of thymol, carvacrol, chorothymol and eugenol against fungi may be attributed to containing the OH group in the aromatic ring. Also, the chlorine atom on the chlorothymol is playing an important role in the fungicidal activity against the tested fungal strains20,23. Therefore, it’s possible to use these monoterpenoids as lead compounds to obtain new friendly fungicides for both humans and the environment.
Fungicidal activity of synthesized monoterpenoidal brominate derivatives against soil and air-born fungi: The fungicidal activity of brominate monoterpenoid derivatives against soil-born fungi i.e., F. oxysporum, R. solani and P. debrinumwas shown in Table (4). The gained results indicated that F. oxysporum was sensitive to treatments of bromocarvacrol (EC50 = 21.86 mg L–1), followed by Bromothymol (EC50 = 26.54 mg L–1), bromoegunol (EC50 = 56.22 mg L–1) and bromochlorothymol (EC50 = 60.10 mg L–1). While Rhizoctonia solani showed high sensitivity towards bromochlorothymol, bromocarvacrol, bromothymol and bromoegunol with EC50 values of 9.38, 10.46, 28.08 and 40.51 mg L–1, consecutively. The fungus P. debrinum was very sensitive to the treatments of bromocarvacrol, bromochlorothymol, bromothymol and bromoegunol with EC50 values of 27.50, 40.93, 47.70 and 165.20 mg L–1, respectively. The standard fungicide (mancozeb) recorded EC50 values of 12.02, 91.70 and 154.60 mg L–1 with F. oxysporum, R. solani and P. debrinum, respectively.
The air-born fungi results clarified that A. alternata was very sensitive to bromocarvacrol, bromothymol and bromochlorothymol with EC50 values of 16.34, 21.88 and 21.94 mg L–1, respectively, while it was less sensitive to bromoegunol (EC50 = 163.69 mg L–1). In the same context, Helminthsporum sp., recorded EC50 values estimated by 12.00, 15.47, 23.30 and 28.82 mg L–1 with bromocarvacrol, bromochlorothymol, bromothymol and bromoegunol, respectively. Botrytis fabae showed sensitivity towards bromothymol (EC50 = 14.80 mg L–1), bromocarvacrol (EC50= 15.36 mg L–1), bromochlorothymol (EC50= 28.11 mg L–1) and bromoegunol (EC50 = 39.04 mg L–1). The treatment of mancozeb recorded EC50 values of 47.44, 115.61 and 236.37 mg L–1 with Helminthsporum sp., A. alternata and B. fabae, respectively. These findings are in agreement with those obtained by Kaur et al.14, who found that halogenated thymol derivatives e.g. chlorothymol, dichlorothymo, monobromothymol and dibromothymol have strong antifungal and antibacterial activities. Also, Chauhan et al.24, found that thymol and its derivatives succeeded to inhibit the mycelia growth of Rhizoctonia solani during the in vitro study, while thesoil application under greenhouse conditions thymol or its derivatives suppressed the damping-off in cucumber seedlings at a range of 26.67 to 100%.
Bactericidal activity of tested monoterpenoids and their brominate derivatives: In the current investigation, the bactericidal activity of four monoterpenoid compounds namely, carvacrol, thymol, chlorothymol and eugenol, in addition to, their brominated derivatives, Bromochlorothymol, Bromothymol, Bromocarvacrol and Bromoeugenol were assessed against A. tumefaciens, E. cartovora, E. amylovora and P. solanacearum under laboratory conditions (Table 5). The bacterium of A. tumefacient was very sensitive to standard bactericide streptomycin with MIC = 5 μg mL–1, followed by E. cartovora, E. amylovora and P. solanacearum with values estimated by 10 μg mL–1 of each.
| Table 4: | Fungicidal activity of tested monoterpenoid derivatives against phytopathogenic fungi | |||
95% confidence limits |
||||
| Fungus | EC50/ppm |
Lower |
Upper |
Slope±SE |
| Bromochlorothymol | ||||
| Fusarium oxysprum | 60.1 |
49.9 |
72.1 |
1.21±0.11 |
| Rhizoctonia solani | 9.38 |
7.8 |
10.9 |
2.30±0.24 |
| Pythium debrinum | 40.93 |
33.4 |
48.6 |
1.97±0.30 |
| Alteranria altranata | 21.94 |
1.25 |
40.3 |
2.00±0.24 |
| Helimanthosporum sp. | 15.47 |
9.4 |
46.8 |
2.50±0.29 |
| Botrytis febae | 28.11 |
15.77 |
50.11 |
1.00±0.10 |
| Bromothymol | ||||
| Fusarium oxysprum | 26.54 |
18.57 |
39.73 |
3.00±0.23 |
| Rhizoctonia solani | 28.08 |
11.54 |
150.56 |
2.30±0.27 |
| Pythium debrinum | 47.7 |
31.07 |
69.17 |
1.30±0.30 |
| Alteranria altranata | 21.88 |
17.17 |
33.35 |
1.40±0.26 |
| Helimanthosporum sp. | 23.3 |
20.26 |
29.5 |
2.80±0.15 |
| Botrytis febae | 14.8 |
9.47 |
23.36 |
2.80±0.23 |
| Bromocarvacrol | ||||
| Fusarium oxysprum | 21.86 |
16.59 |
27.44 |
1.63±0.15 |
| Rhizoctonia solani | 10.46 |
9.14 |
12.23 |
2.55±0.4 |
| Pythium debrinum | 27.5 |
23.29 |
33.626 |
1.82±0.19 |
| Alteranria altranata | 16.34 |
14.42 |
18.57 |
2.25±0.2 |
| Helimanthosporum sp. | 12 |
10.4 |
14.6 |
2.40±0.4 |
| Botrytis febae | 15.36 |
12.64 |
18.71 |
4.00±0.31 |
| Bromoeugenol | ||||
| Fusarium oxysprum | 56.22 |
28.7 |
70.2 |
2.70±0.25 |
| Rhizoctonia solani | 40.51 |
18.78 |
61.32 |
2.00±0.19 |
| Pythium debrinum | 165.2 |
139 |
200.2 |
1.70±0.23 |
| Alteranria altranata | 163.69 |
138.32 |
198.3 |
1.70±0.23 |
| Helimanthosporum sp. | 28.82 |
11.81 |
51.42 |
1.14±0.11 |
| Botrytis febae | 39.04 |
20.88 |
61.65 |
1.30±0.12 |
| Mancozeb | ||||
| Fusarium oxysprum | 12.02 |
4.59 |
25.82 |
1.38±0.06 |
| Rhizoctonia solani | 154.6 |
124.82 |
192.62 |
1.18±0.11 |
| Pythium debrinum | 91.7 |
61.93 |
294.8 |
0.90±0.20 |
| Alteranria altranata | 115.61 |
77.65 |
170.97 |
0.62±0.09 |
| Helimanthosporum sp. | 47.44 |
30.83 |
56.37 |
0.77±0.09 |
| Botrytis febae | 236.37 |
127.53 |
612.23 |
1.35±0.1 |
| SE: Standard error | ||||
| Table 5: | Bactericidal activity of some monoterpenoids and their derivatives by minimal inhibitory concentration (MIC) | |||
| Compound (μg mL–1) | A. tumefaciens |
E. amylovora |
E. cartovora |
P. solanacearm |
| Chlorothymol | 35 |
50 |
35 |
60 |
| Thymol | 100 |
90 |
300 |
>300 |
| Eugenol | 220 |
300 |
>300 |
300 |
| Carvacrol | 45 |
50 |
50 |
50 |
| Bromochlorothymol | 5 |
50 |
5 |
60 |
| Bromothymol | 9 |
200 |
10 |
3 |
| Bromoeugenol | 5 |
50 |
5 |
5 |
| Bromocarvacrol | 5 |
5 |
5 |
5 |
| Sterptomycin | 5 |
10 |
10 |
10 |
With respect to the A. tumefaciens strain it was sensitive to chlorothymol, carvacrol, thymol and eugenol with values of 35, 45, 100 and 220 μg mL–1, respectively. Meanwhile, A. tumefaciens showed great sensitivity towards bromochlorothymol, bromocarvacrol, bromoeugenol and bromothymol with MIC values of 5, 5, 5 and 9 μg mL–1, respectively. However, E. amylovora strain exhibited sensitivity to chlorothymol, carvacrol, thymol and eugenol with MIC values of 50, 50, 50 and 300 μg mL–1, respectively. Whilst, E. amylovora recorded sensitivity towards bromocarvacrol (5 μg mL–1), bromochlorothymol (50 μg mL–1), bromoeugenol (50 μg mL–1) and bromothymol (200 μg mL–1).
On the other hand, E. cartovora recorded sensitivity towards tested monoterpenoids such as chlorothymol, carvacrol, thymol and eugenol with values of 35, 50, 300 and >300 μg mL–1, respectively. However, E. cartovora exhibited high sensitivity to bromochlorothymol (5 μg mL–1), bromocarvacrol (5 μg mL–1), bromoeugenol (5 μg mL–1) and bromothymol (10 μg mL–1). According to the obtained data, P. solanacearum showed sensitivity towards carvacrol, chlorothymol, eugenol and thymol with MIC values estimated by 50, 60, 300 and >300, consecutively. Furthermore, the transformed monoterpenoids such as bromothymol, bromocarvacrol, bromoeugenol and bromochlorothymol were very effective against P. solanacearum achieving 3, 5, 5 and 60 μg mL–1, respectively.
Several studies reported that the monoterpenes exhibited antifungal and antibacterial activities against a wide range of microorganisms25-28. The results of the current investigation are in agreement with El-Zemity et al.29 who stated that chlorothymol was effective as a bactericide against Agrobacterium tumefaciens and Erwinia carotovora var carotovora. The activity of chlorothymol may be attributed to the chlorine atom which plays a good role in the bactericidal activity against the tested bacteria. Also, Mahboub and Memmou30 found that synthesized 6-bromoeugenol or eugenol has antibacterial activity, especially in both positive and negative gram bacteria. Also, thymol derivatives had antibacterial activities with minimum inhibitory concentration values, 40 to 80μg mL–1 31. Thymyl-4-nitrobenzoate recorded good inhibitory action towards plant pathogenic bacteria either as a major constituent or in combination with other antimicrobial agents32. Abd-El-Aziz et al.33 indicated that using the essential oils of caraway (Carum carvi L.) and thyme (Thymus vulgaris L.) exhibited antibacterial efficacy against Agrobacterium tumefaciens in a laboratory study. Moreover, the application of caraway and thyme oils suppressed the gall formation by 63.16 and 89.47%, respectively, on apricot seedlings in pots.
CONCLUSION
From the above results, it could be concluded that chlorothymol (monoterpene) recorded the highest fungicidal activity against the most of soil and air-born fungi. While, the brominated derivatives of monoterpenoids showed that bromocarvacrol was the most effective against all tested fungi, whereas, bromoegunol was the least one. The same trend was recorded with bacteria, where chlorothymol (monoterpene) recorded the highest bactericidal activity. However, the brominated derivatives of monoterpenoids were more effective against all the tested bacteria. Therefore, this study proposed some eco-friendly solutions, but more experiments are needed in the future.
SIGNIFICANCE STATEMENT
Fungi and bacteria are among the most common plant pathogens that lead to losses in agricultural crops, whether during planting or after harvest. Combating these diseases has become costly, especially recently, so it was necessary to search for effective alternatives that can be used safely without any adverse effects. This study was thus conducted to assess the impact of some monoterpenes and their brominate derivatives against soil and air fungi as well as bacteria. Also, results showed that some of the monoterpenes or their derivatives e.g., chlorothymol and bromocarvacrol were effective as fungicides and/or bactericides.
REFERENCES
- Chandler, D., A.S. Bailey, G.M. Tatchell, G. Davidson, J. Greaves and W.P. Grant, 2011. The development, regulation and use of biopesticides for integrated pest management. Philos. Trans. R. Soc. B: Biol. Sci., 366: 1987-1998.
- Heydari, A. and M. Pessarakli, 2010. A review on biological control of fungal plant pathogens using microbial antagonists. J. Biol. Sci., 10: 273-290.
- Omezzine, F., M. Bouaziz, M. Daami-Remadi, M.S.J. Simmonds and R. Haouala, 2017. Chemical composition and antifungal activity of Trigonella foenum-graecum L. varied with plant ploidy level and developmental stage. Arabian J. Chem., 10: S3622-S3631.
- Shuping, D.S.S. and J.N. Eloff, 2017. The use of plants to protect plants and food against fungal pathogens: A review. Afr J. Tradit. Complementary Altern. Med., 14: 120-127.
- Silvan, J.M., E. Guerrero-Hurtado, A. Gutierrez-Docio, M. Prodanov and A.J. Martinez-Rodriguez, 2022. Olive leaf as a source of antibacterial compounds active against antibiotic-resistant strains of Campylobacter jejuni and Campylobacter coli. Antibiotics, 12: 26.
- Chodkowska, K.A., H. Iwiński, K. Wódz, T. Nowak and H. Różański, 2022. In vitro assessment of antimicrobial activity of phytobiotics composition towards of avian pathogenic Escherichia coli (APEC) and other E. coli strains isolated from broiler chickens. Antibiotics, 11: 1818.
- Vaou, N., E. Stavropoulou, C. Voidarou, C. Tsigalou and E. Bezirtzoglou, 2021. Towards advances in medicinal plant antimicrobial activity: A review study on challenges and future perspectives. Microorganisms, 9: 2041.
- Zielińska-Błajet, M. and J. Feder-Kubis, 2020. Monoterpenes and their derivatives-recent development in biological and medical applications. Int. J. Mol. Sci., 21: 7078.
- Stephane, F.F.Y. and B.K.J. Jules, 2020. Terpenoids as Important Bioactive Constituents of Essential Oils. In: Essential Oils - Bioactive Compounds, New Perspectives and Applications, de Oliveira, M.S., S. Silva and W.A. da Costa (Eds.), IntechOpen, London, UK, ISBN: 978-1-83962-698-2, pp: 222.
- Marinelli, L., A. di Stefano and I. Cacciatore, 2018. Carvacrol and its derivatives as antibacterial agents. Phytochem. Rev., 17: 903-921.
- Wang, Y., J. Ren, X.H.H. Huang, R. Tong and J.Z. Yu, 2017. Synthesis of four monoterpene-derived organosulfates and their quantification in atmospheric aerosol samples. Environ. Sci. Technol., 51: 6791-6801.
- Wang, K., S. Jiang, T. Pu, L. Fan, F. Su and M. Ye, 2019. Antifungal activity of phenolic monoterpenes and structure-related compounds against plant pathogenic fungi. Nat. Prod. Res., 33: 1423-1430.
- Cui, Z., X. Li and Y. Nishida, 2014. Synthesis and bioactivity of novel carvacrol and thymol derivatives containing 5-phenyl-2-furan. Lett. Drug Des. Discovery, 11: 877-885.
- Kaur, R., M.P. Darokar, S.K. Chattopadhyay, V. Krishna and A. Ahmad, 2014. Synthesis of halogenated derivatives of thymol and their antimicrobial activities. Med. Chem. Res., 23: 2212-2217.
- Selim, R.E., S.M. Ahmed, S.R. El-Zemity, S.S. Ramses and Y.T.A. Moustafa, 2015. Antifungal activity and seasonal variation of green alga (Ulva lactuca) extracts. Asian J. Agric. Food Sci., 3: 419-427.
- Tian, L., X. Chen, X. Jia, S. Wang and X. Wang et al., 2021. First report of antifungal activity conferred by non-conventional peptides. Plant Biotechnol. J., 19: 2147-2149.
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID), 2000. Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by agar dilution. Clin. Microb. Infect., 6: 509-515.
- Finney, D.J., 1971. Probit Analysis. 3rd Edn., Cambridge University Press, Cambridge, ISBN: 9780521080415, Pages: 333.
- Numpaque, M.A., L.A. Oviedo, J.H. Gil, C.M. García and D.L. Durango, 2011. Thymol and carvacrol: Biotransformation and antifungal activity against the plant pathogenic fungi Colletotrichum acutatum and Botryodiplodia theobromae. Trop. Plant Pathol., 36: 3-13.
- Kordali, S., A. Cakir, H. Ozer, R. Cakmakci, M. Kesdek and E. Mete, 2008. Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresour. Technol., 99: 8788-8795.
- Lee, O.G., G.J. Choi, K.S. Jang, H.K. Lim, K.Y. Cho and J.C. Kim, 2007. Antifungal activity of five plant essential oils as fumigant against postharvest and soilborne plant pathogenic fungi. Plant Pathol. J., 23: 97-102.
- Tsao, R. and T. Zhou, 2000. Antifungal activity of monoterpenoids against postharvest pathogens Botrytis cinerea and Monilinia fructicola. J. Essent. Oil Res., 12: 113-121.
- Abbaszadeh, S., A. Sharifzadeh, H. Shokri, A.R. Khosravi and A. Abbaszadeh, 2014. Antifungal efficacy of thymol, carvacrol, eugenol and menthol as alternative agents to control the growth of food-relevant fungi. J. Mycol. Med., 24: e51-e56.
- Chauhan, K.R., T.C. Le, P.K. Chintakunta and D.K. Lakshman, 2017. Phyto-fungicides: Structure activity relationships of the thymol derivatives against Rhizoctonia solani. J. Agric. Chem. Environ., 6: 175-185.
- Marchese, A., I.E. Orhan, M. Daglia, R. Barbieri and A.D. Lorenzo et al., 2016. Antibacterial and antifungal activities of thymol: A brief review of the literature. Food Chem., 210: 402-414.
- de Castro Teixeira, A.P., R. de Oliveira Nóbrega, E. de Oliveira Lima, W. de Oliveira Araújo and I. de Oliveira Lima, 2020. Antifungal activity study of the monoterpene thymol against Cryptococcus neoformans. Nat. Prod. Res., 34: 2630-2633.
- Zhou, L., Z. Zhang, M. Wei, Y. Xie, S. He, H. Shi and Z. Lin, 2019. Evaluation of the antifungal activity of individual and combined monoterpenes against Rhizopus stolonifer and Absidia coerulea. Environ. Sci. Pollut. Res., 26: 7804-7809.
- Ahmed, B., A. Jailani, J.H. Lee and J. Lee, 2022. Inhibition of growth, biofilm formation, virulence, and surface attachment of Agrobacterium tumefaciens by cinnamaldehyde derivatives. Front. Microbiol., 13: 1001865.
- El-Zemity, S.R., M.A. Radwan, S.A.E.M. Mohamed and S.M. Sherby, 2008. Antibacterial screening of some essential oils, monoterpenoids and novel N-methyl carbamates based on monoterpenoids against Agrobacterium tumefaciens and Erwinia carotovora. Arch. Phytopathol. Plant Protect., 41: 451-461.
- Mahboub, R. and F. Memmou, 2016. Antimicrobial properties of 6-bromoeugenol and eugenol. Int. Lett. Nat. Sci., 53: 57-64.
- Swain, S.S., S.K. Paidesetty and R.N. Padhy, 2019. Synthesis of novel thymol derivatives against MRSA and ESBL producing pathogenic bacteria. Nat. Prod. Res., 33: 3181-3189.
- Kumar, A., S.P. Singh and S.S. Chhokar, 2008. Thymol and its derivatives as antimicrobial agents. Nat. Prod. Commun., 3.
- Abd-El-Aziz, R.M., A.F. Abd El-Rahman and D.M.G. Hendi, 2021. Control of Agrobacterium tumefaciens with essential oils compared to antagonistic Agrobacterium radiobacter strain K84. Egypt. J. Phytopathol., 49: 80-92.
How to Cite this paper?
APA-7 Style
Ahmed,
S.M., Selim,
R.E., Khalil,
M.S., El-Zemity,
S.R. (2023). Monoterpenoids and Their Synthesized Brominate Derivatives as Eco-Friendly Measures to Control Some Plant Pathogenic Fungi and Bacteria. Asian Journal of Biological Sciences, 16(3), 264-274. https://doi.org/10.3923/ajbs.2023.264.274
ACS Style
Ahmed,
S.M.; Selim,
R.E.; Khalil,
M.S.; El-Zemity,
S.R. Monoterpenoids and Their Synthesized Brominate Derivatives as Eco-Friendly Measures to Control Some Plant Pathogenic Fungi and Bacteria. Asian J. Biol. Sci 2023, 16, 264-274. https://doi.org/10.3923/ajbs.2023.264.274
AMA Style
Ahmed
SM, Selim
RE, Khalil
MS, El-Zemity
SR. Monoterpenoids and Their Synthesized Brominate Derivatives as Eco-Friendly Measures to Control Some Plant Pathogenic Fungi and Bacteria. Asian Journal of Biological Sciences. 2023; 16(3): 264-274. https://doi.org/10.3923/ajbs.2023.264.274
Chicago/Turabian Style
Ahmed, Soad, Mohamedeen, Rasha El-Sayed Selim, Mohamed Salah Khalil, and Saad Rashad El-Zemity.
2023. "Monoterpenoids and Their Synthesized Brominate Derivatives as Eco-Friendly Measures to Control Some Plant Pathogenic Fungi and Bacteria" Asian Journal of Biological Sciences 16, no. 3: 264-274. https://doi.org/10.3923/ajbs.2023.264.274

This work is licensed under a Creative Commons Attribution 4.0 International License.



