Evaluation of Phytochemical Constituents and Elemental Profiling of Selected Medicinal Plants in South-West, Nigeria
| Received 25 Dec, 2023 |
Accepted 21 Feb, 2024 |
Published 31 Mar, 2024 |
Background and Objective: Phytochemicals are responsible for the definite physiological effects exerted on the human body by various parts of the plant and it has been reported to play a major role in nutrition and control of diseases. This study aimed to comparatively investigate the phytochemical and elemental components of five medicinal plants commonly used in South-Western, Nigeria. Materials and Methods: Qualitative and quantitative phytochemical screening of all the extracts was done using different standard methods. Calcium, potassium, magnesium, phosphorus, iron and other elements were determined using standard procedures. Results: Preliminary qualitative phytochemical screening indicates the presence of glycosides, saponins, tannins, flavonoids, alkaloids and anthraquinones. The quantitative phytochemical screening revealed that glycosides, saponins, tannins, flavonoids, alkaloids and anthraquinones were significantly (p<0.05) higher in ethanol extract compared to ethyl acetate extract in all the studied medicinal plants, however, the reverse was the case in Urena lobata. Sodium (Na), potassium (K) and calcium (Ca) were significantly (p<0.05) higher in ethyl acetate extract than in ethanol extract in all the studied medicinal plants. While, iron (Fe), lead (Pb), cadmium (Cd) and arsenic (As) were significantly (p<0.05) higher in ethanol extract compared to ethyl acetate extract in all the studied medicinal plants. Conclusion: This study suggests the different range of phytochemicals present in the medicinal plants studied with their potential therapeutic benefits, emphasizing the significance of these plants in traditional and modern medicine. Furthermore, the elemental composition sheds light on the nutritional and essential value of these plants, which could contribute to their holistic healing properties.
| Copyright © 2024 Usin et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Medicinal plants produce numerous secondary metabolites, including bioactive compounds like alkaloids, tannins, flavonoids, sterols and terpenes1. These compounds are essential for drug efficacy2 and are responsible for the physiological effects exerted by plant parts on the human body3. They play a significant role in nutrition and disease control1. Bioactive compounds, also known as phytochemicals, are widely used in herbal medicines worldwide for treating various health challenges4.
Citrus sinensis, also known as sweet oranges, belongs to the family Rutaceae. It is a native of Asia and is now widespread globally5. It is a rich source of vitamin C, a powerful antioxidant that builds the body's immune system6. Citrus sinensis is traditionally used to treat various ailments, including cramps, colic, stress, depression, hypertension, cold, cough, obesity, angina and tuberculosis7. It has been shown to prevent diseases like cancer, cardiovascular disease, neurological diseases and the ageing process. Its antibacterial, antifungal, antiparasitic, antioxidant, hypocholesterolemic, anti-obesity, antiosteoporotic, relaxant, sedative and anxiolytic activities have been reported8-11.
Lawsonia inermis, also known as henna, mignonette and Egyptian privet, is a shrub with reddish-brown bark and opposite, sub-sessile, elliptic or broadly lanceolate leaves12. It is commonly grown in Africa, the Middle East and India13. It belongs to the Lythraceae family and has astringent, anti-hemorrhagic, hypotensive, cardio-inhibitory, sedative, hypoglycemic, immunostimulant, hepatoprotective, anti-inflammatory, tuberculostatic, anti-cancer and antioxidant properties14-16. The plant is used for various health conditions, including rheumatoid arthritis, headaches, ulcers, diarrhoea, fever, heart disease, hair loss, leukorrhea, smallpox, skin disease, burns and diabetes17. The primary chemical components of henna are Lawsone (2-hydroxynaphthoquinone), mucilage, mannite, gallic acid and tannic acid15. The plant also contains coumarins, flavonoids, naphthalene, derivatives of gallic acid, triterpenoids, steroids and aliphatic hydrocarbons13.
Albizia zygia L., which belongs to the family Fabaceae. It is also known as West African walnut. It is indigenous to West and East Africa, India and Australia18. In traditional medicine, Albizia zygia is used to cure several ailments, including fever, malaria, diarrhoea, oedema, insanity, wounds, toothaches, yaws, sores, waist pain, fever and venereal diseases19,20. Albizia zygia exhibits cytotoxicity against human T-lymphoblast-like leukaemia and prostate and breast cancer cell lines21. Phytochemical studies reveal that the plant contains cardiovascular glycosides, flavonoids, tannins and triterpenes22. Some reported pharmacological activities of A. zygia include antiprotozoal19, antimalarial23, gastro-protective24, anti-inflammatory and anti-nociceptive20,25, anti-depressant and anti-psychotic26,27, antimicrobial and antioxidant28, antipyretic and analgesic29.
Urena lobata L., belongs to the family Malvaceae. It is commonly known as Caesar weed or Congo jute. It is grown in Brazil, Southeast Asia and the Congo and widely distributed throughout the world30. Urena lobata is a well-known plant in folk medicine used in the treatment of diuretic, febrifuge, dysentery, cough, dropsy, edema, abdominal colic, rheumatism, leucorrhea and gonorrhea31, due to its wide range of biological activities, which include anti-hyperglycemic, antioxidant, anti-nociceptive, anti-diarrheal, anti-inflammatory, anti-proliferative, antibacterial, anxiolytic and wound healing activities32-34. A phytochemical screening of the plant revealed the presence of mangiferin, quercetin, flavonoids, lignans, coumarins, triglycerides, phenylethyl glycosides, megastigmane glycosides, lignins and urenalignosides A-D33,35-38.
Olax subscorpioidea is a tree or shrub that grows widely in Nigeria, Zaire, Senegal and other regions of Africa, which belongs to the family of Olacaceae39. Traditionally, in Nigeria, it is one of the recipes used to treat cancer and asthma40. More so, it is used as a chewing stick for oro-dental hygiene, treatment of inflammatory and mental diseases, convulsion, pain, obesity, diabetes mellitus, malaria, parasitic infections, diarrhoea, gastric ulcer, depression, fever, anxiety and constipation40-43. In Congo Republic, the decoction of the stem, bark and leaves of O. subscorpioidea has been used against rheumatism and arthritis pains40, venereal diseases and guinea worms44. Preliminary phytochemical screening revealed the presence of saponins, alkaloids, tannins, phenols, phlobatannins, carbohydrates, cardiac glycosides, terpenoids, steroids and flavonoids. These phytochemicals are responsible for the analgesic properties39,40. Also, elemental studies revealed the presence of calcium, copper, manganese, magnesium, sodium, zinc, potassium, aluminium, silicon, sulphur, cobalt, nickel, bromine, phosphorus, chlorine, iron, rubidium and strontium45,46. Pharmacological activities include antidepressant, anticonvulsant, sedative, anxiolytic, anti-Alzheimer’s, cytotoxicity, antioxidant, hypolipidemic, analgesic, anti-inflammatory, antiarthritic, hypoglycaemic, antiulcer, anticancer, antimicrobial, anthelmintic, hepatoprotective, apoptotic and antiprotease activities39,40,45,46.
It is well-known that plants produce phytochemicals to protect themselves, however, it has been demonstrated that they can also be used to protect humans against numerous diseases47. Globally, there are more than a thousand known phytochemicals48,49. Some phytochemicals known as phytotoxins are toxic to humans50, for instance, aristolochic acid is carcinogenic even at low doses51, while some phytochemicals are anti-nutrients, i.e. they interfere with the absorption of nutrients. Others, such as some polyphenols and flavonoids, may be pro-oxidants in high ingested amounts52. Minerals play vital metabolic and physiological roles in the living system53. They are also regarded as activators, as well as components of many plant enzymes54. Deficiency as well as excess amount/quantity of the minerals might disturb normal biochemical functions of the body leading to abnormal physiological functions too55. Therefore, this study seeks to comparatively investigate the phytochemicals (quantitative and qualitative) and mineral elements of ethyl acetate and ethanol extracts of these medicinal plants commonly used by the Yoruba tribe in South-Western Nigeria for cosmetics, food and medicinal purposes.
MATERIALS AND METHODS
Study area: The study was designed and carried out in the Department of Science Laboratory Technology, School of Sciences and Engineering, D. S. Adegbenro ICT Polytechnic, Ekuru-Itori, Ewekoro, Ogun State, Nigeria. The duration was from October, 2021 to December, 2021.
Collection and identification of plant materials: Fresh leaves of Lawsonia Inermis, Albizia zygia, Citrus sinensis, Urena lobata and Olax subscorpioidea were collected from different locations within Ewekoro Local Government Area in Ogun State and Afijio Local Government Area in Oyo State, Nigeria. The identification and authentication of the plant materials were done at Forest Research of Nigeria, Jericho Hill, Ibadan, Oyo State, with the voucher number FHI 113335 (Olax subscorpioidea), FHI 113336 (Lawsonia Inermis), FHI 113355 (Urena lobata), FHI 113356 (Albizia zygia) and FHI 113360 (Citrus sinensis) deposited for further reference.
Extraction of plant materials: The collected samples were air-dried at room temperature for 21 days. The samples were ground into powder using a pulverizer and stored in an air-tight bottle. After weighing, 200 g of the ground sample of the leaf was dissolved in 500 mL each of ethanol and ethyl acetate and was kept in the refrigerator for 72 hrs with intermittent shaking. The extract was filtered using a chess cloth and Whatman filter paper No. 1 (125 mm), to obtain filtrates of the respective solvents of ethanol and ethyl acetate. The filtrates were evaporated to dryness using a water bath (at 40°C) to obtain the slurry, which was persevered in a phial, labelled appropriately, stored in the refrigerator at 4°C and was used for phytochemical and mineral analysis.
Preliminary qualitative screening of phytochemical constituents: Preliminary qualitative analysis of phytochemical constituents was carried out to identify the secondary metabolites present in the ethyl acetate and ethanol extracts of Lawsonia inermis, Albizia zygia, Citrus sinensis, Urena lobata and Olax subscorpioidea leaves56,57. The qualitative analysis of the various phytochemicals was carried out by using Meyer’s reagents (alkaloids). Other tests carried out include the Frothing test (saponins), ferric chloride test (tannins), Schinoda test (flavonoids), Fehling’s test (glycosides) and Borntrager’s test (anthraquinones).
Quantitative screening of phytochemical constituents
Determination of alkaloid: Alkaloid was determined according to the method of Obadoni and Ochuko58 and the percentage of total alkaloid content was calculated as:
Determination of flavonoid: The total flavonoid content was determined using the procedure described by Ejikeme et al.59. The percentage of total flavonoid content was calculated as:
 |
Determination of saponin: Saponin quantitative determination was carried out using the method described by Obadoni and Ochuko58. The saponin content is calculated as percentage:
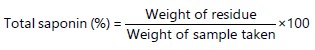 |
Determination of tannin: Tannin content was determined using the method of Kaur and Arora60, using tannic acid as standard. The percentage tannin content was calculated using the formula:
 |
Determination of glycoside: Glycoside content was determined using the method of Ezeonu and Ejikeme61. The glycoside content is calculated as percentage using the formula:
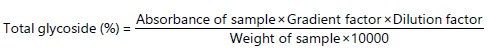 |
Determination of anthraquinones: Anthraquinones was determined using the method described by Bussmann et al.62. The percentage anthraquinone is calculated using the formula:
Determination of the mineral composition: The mineral contents of the plants were determined by the procedure of Adelantado et al.63. Calcium, potassium, magnesium, phosphorus, iron and other elements were measured with Atomic Absorption Spectrophotometer (ThermoFisher Scientific S Series Model GE 712354) after digesting with perchloric-nitric acid mixture. The extracts were digested and placed in a 125 mL Erlenmeyer flask. Perchloric acid (4 mL), concentrated sulphuric acid (2 mL) and concentrated HNO3 (25 mL) were added and the mixture was heated in a digester until a thick white fume appeared. The heating process was sustained for 30 sec, then cooled. About 50 mL of distilled water was added and the solution was filtered and diluted with distilled water into a Pyrex volumetric flask (564050/EMD; FisherScientific Inc., Pittsburgh, Pennsylvania, USA). The process was repeated until the solution was a consistent and effective solvent. Subsequently, the mixture wasanalyzedutilizing an Atomic Absorption Spectrophotometer (ThermoFisher Scientific Inc., Waltham, Massachusetts, USA).
Statistical analysis: Statistical analysis was carried out using One-way Analysis of Variance (ANOVA) test. Resulting data were represented as Mean±Standard Deviation of triplicate determinations. GraphPad Prism 9® (Version 9.3.1, GraphPad Software Inc., San Diego, United States of America) software was used for the statistical analysis and statistical significance was obtained at p<0.05.
RESULTS AND DISCUSSION
It is no doubt that most plants have at least a medicinal property, which is because of secondary metabolites present in such plants. In this study, plants such as Lawsonia inermis, Albizia zygia, Citrus sinensis, Urena lobate and Olax subscorpioidea were considered and analysed to elucidate its various phytochemicals present and comparatively evaluate which solvents yield more of the bioactive constituents. The qualitative phytochemical screening indicates the presence of glycosides, saponins, tannins, flavonoids, alkaloids and anthraquinones (Table 1).
The quantitative phytochemical screening revealed that glycosides, saponins, tannins, flavonoids, alkaloids and anthraquinones were significantly (p<0.05) higher in ethanol extract compared to ethyl acetate extract in all the studied medicinal plants, however, the reverse was the case in Urena lobata (Table 2). Comparatively, tannin content was higher in Urena lobata in both solvents, followed by Lawsonia inermis, Citrus sinensis, Olax subscorpioidea and Albizia zygia in descending order. Saponin content was higher in Lawsonia inermis in ethyl acetate solvent, followed by Urena lobata, Citrus sinensis, Olax subscorpioidea and Albizia zygia, but for the ethanol solvent, Lawsonia inermis was still the highest followed by Urena lobata, Olax subscorpioidea, Citrus sinensis and Albizia zygia. Alkaloid content was higher in Lawsonia inermis in both solvents, followed by Urena lobata, Olax subscorpioidea, Citrus sinensis and Albizia zygia. Glycoside content was higher in Urena lobata in both solvents, followed by Lawsonia inermis, Citrus sinensis, Olax subscorpioidea and Albizia zygia. Flavonoid content was higher in Citrus sinensis in ethyl acetate solvent, followed by Lawsonia inermis, Urena lobata, Albizia zygia and Olax subscorpioidea. For the ethanol solvent, Citrus sinensis was higher followed by Lawsonia inermis, Albizia zygia, Olax subscorpioidea and Urena lobata. Anthraquinone content was higher in Urena lobata in ethyl acetate solvent, followed by Albizia zygia, Lawsonia inermis, Citrus sinensis and Olax subscorpioidea, but for the ethanol solvent, Albizia zygia has the highest content followed by Urena lobata, Citrus sinensis, Lawsonia inermis and Olax subscorpioidea.
The results of this investigation corroborated the numerous phytochemical investigations conducted on these plants8,13,22,33,35-40. Phytochemicals provide medical benefits to human beings. For example, saponin has demonstrated promising medicinal capabilities in the treatment of many conditions such as cardiovascular diseases, infections from bacteria, cancer and diabetes64,65. Flavonoids have been acknowledged for their advantageous impact on wellness. They are also considered an essential element in multiple pharmacological, nutritional product, therapeutic and cosmetic applications66. This compound possesses anti-oxidative, anti-mutagenic, anti-inflammatory and anti-carcinogenic effects, as well as the capacity to regulate essential cellular enzyme efficiency66.
Research has shown that the phytochemicals found in plants can potentially be influenced by the extraction procedures employed and the specific formulations of solvents used65, so the concentration of such phytochemicals is determined by the state of the chemical employed as the solvents in the extraction method, in addition to its development and preservation situations67,68 However, considering the various factors that can influence the identification of these chemicals in plants, it is unsurprising that certain compounds were found in significant quantities in the ethyl acetate solvent, while others were present in the ethanol solvent.
It is vital to know that the therapeutic benefits of herbal remedies in conventional medicine can be linked to the presence of trace elements in extremely small amounts. Essential trace elements, like iron, play a dual role in both treating and preventing disorders such as anaemia69. The elemental analysis revealed the presence of sodium (Na), potassium (K), calcium (Ca), iron (Fe), lead (Pb), cadmium (Cd) and arsenic (As). The result shows that sodium (Na), potassium (K) and calcium (Ca) were present in higher amounts in ethyl acetate extract than in ethanol extract in all the studied medicinal plants. While, iron (Fe), lead (Pb), cadmium (Cd) and arsenic (As) were present in high amount in ethanol extract compared to ethyl acetate extract in all the studied medicinal plants (Table 3).
| Table 1: | Qualitative phytochemical analysis of ethyl acetate and ethanol extracts of some medicinal plants commonly in South-Western, Nigeria | |||
Lawsonia inermis |
Abizia zygia |
Citrus sinensis |
Urena lobata |
Olax subscorpiodea |
|||||||
| Phytochemicals | Tests used | Ethyl acetate |
Ethanol |
Ethyl acetate |
Ethanol |
Ethyl acetate |
Ethanol |
Ethyl acetate |
Ethanol |
Ethyl acetate |
Ethanol |
| Tannins | Ferric chloride test | ++ |
++ |
+ |
+ |
+ |
+ |
++ |
++ |
+ |
+ |
| Saponins | Frothing test | ++ |
++ |
+ |
+ |
+ |
+ |
++ |
+ |
+ |
+ |
| Alkaloids | Meyer’s reagents | ++ |
+++ |
+ |
+ |
+ |
++ |
+++ |
++ |
++ |
++ |
| Flavonoids | Schinoda test | ++ |
++ |
++ |
++ |
++ |
+++ |
++ |
++ |
+ |
++ |
| Glycoside | Fehling’s test | + |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
| Anthraquinone | Borntrager’s test | + |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
| +: Present, ++: Present in moderate and +++: Present in more quantity | |||||||||||
| Table 2: | Quantitative phytochemical analysis of ethyl acetate and ethanol extracts of some medicinal plants commonly in South-Western, Nigeria (%) | |||
Lawsonia inermis |
Abizia zygia |
Citrus sinensis |
Urena lobata |
Olax subscorpiodea |
||||||
| Phytochemicals | Ethyl acetate | Ethanol | Ethyl acetate | Ethanol | Ethyl acetate | Ethanol | Ethyl acetate | Ethanol | Ethyl acetate | Ethanol |
| Tannins | 5.12±0.104a | 6.32±0.280b | 1.32±0.120c | 1.58±0.060cd | 3.13±0.050e | 3.51±0.105f | 8.13±0.040g | 7.32±0.100 h | 1.32±0.120cd | 2.89±0.100e |
| Saponins | 6.92±1.00a | 8.14±0.860a | 1.82±0.020b | 2.11±0.010bc | 3.48±0.030cd | 2.55±0.100bcde | 5.66±0.730a | 3.51±0.105cdef | 2.11±0.110bdef | 3.76±0.223cdef |
| Alkaloids | 8.93±1.030a | 12.18±0.920b | 4.36±1.000c | 4.16±0.071cd | 4.82±0.200cde | 6.91±0.010af | 10.67±1.050abg | 8.91±1.010afgh | 5.89±0.300cdef | 8.14±0.140afh |
| Flavonoids | 7.38±1.120a | 9.11±0.980ab | 6.92±1.030abc | 6.11±1.110ad | 8.66±1.000abcde | 10.45±1.050be | 7.61±0.890abcdef | 5.92±0.980acdefg | 3.72±0.120dg | 6.62±1.000abcdefg |
| Glycoside | 0.84±0.040a | 1.02±0.020b | 0.44±0.050c | 0.38±0.017cd | 0.61±0.020e | 0.78±0.010af | 1.21±0.021g | 1.16±0.010 g | 0.45±0.010cd | 0.78±0.010af |
| Anthraquinone | 0.57±0.092a | 0.61±0.021ab | 0.810.030c | 0.71±0.015bcd | 0.41±0.030e | 0.67±0.080abdf | 0.83±0.017cd | 0.69±0.020abcdf | 0.28±0.010g | 0.52± 0.030abe |
| Values were performed in triplicates and represented as Means±Standard Deviations, identical superscript (i.e. a) on the same row means there is no significant difference between the comparing groups at p>0.05 and non-identical superscripts (i.e. a, b, c, d, e, f, g, h) on the same row means there is significance between the comparing groups at p<0.05 | ||||||||||
| Table 3: | Mineral compositions of ethyl acetate and ethanol extracts of some medicinal plants commonly in South-Western, Nigeria (mg/L) | |||
Lawsonia inermis |
Abizia zygia |
Citrus sinensis |
Urena lobata |
Olax subscorpiodea |
||||||
| Minerals | Ethyl acetate | Ethanol | Ethyl acetate | Ethanol | Ethyl acetate | Ethanol | Ethyl acetate | Ethanol | Ethyl acetate | Ethanol |
| Sodium (Na) | 4.17±0.100a | 3.65±0.173b | 3.82±0.112b | 3.12±0.016c | 3.11±0.014c | 2.65±0.004d | 2.60±0.004d | 2.14±0.003e | 2.87±0.001f | 2.34±0.002e |
| Potassium (K) | 1882.11±1.000a | 1369.28±1.000b | 1569.14±1.000c | 1302.45±2.000d | 2345.18±1.000e | 1894.10±4.000f | 1873.41±2.000g | 1518.34±2.000h | 2156.384±2.000i | 1821.635±1.000j |
| Calcium (Ca) | 81.61±1.000a | 77.34±1.732b | 75.62±1.000bc | 70.07±0.9663d | 78.09±1.000bc | 73.59±1.000c | 60.78±1.000e | 52.89±1.004f | 65.38±1.005g | 61.09±1.000h |
| Iron (Fe) | 172.69±2.100a | 181.16±1.528b | 211.68±1.000c | 228.91±3.000d | 181.796±1.000be | 185.67±2.000bef | 178.01±2.000be | 184.50±1.001bef | 206.78±1.000c | 219.00±1.008g |
| Lead (Pb) | 0.038±0.002a | 0.132±0.002b | 0.098±0.002c | 0.176±0.003d | 0.112±0.002e | 0.172±0.001d | 0.142±0.001f | 0.193±0.002g | 0.211±0.001h | 0.284±0.002i |
| Cadmium (Cd) | 0.148±0.001a | 0.213±0.001b | 0.288±0.001c | 0.314±0.002d | 0.442±0.002e | 0.496±0.001f | 0.384±0.004g | 0.411±0.001h | 0.561±0.001i | 0.602±0.002j |
| Arsenic (As) | 0.811±0.002a | 0.981±0.003b | 0.653±0.003c | 0.783±0.097ad | 0.448±0.052e | 0.634±0.034cf | 0.602±0.004cfg | 0.814±0.004ad | 0.416±0.006ef | 0.573±0.003cg |
| Values were performed in triplicates and represented as Means±Standard Deviations, identical superscript (i.e., a) on the same row means there is no significant difference between the comparing groups at p>0.05 and non-identical superscripts (i.e., a, b, c, d, e, f, g, h, i) on the same row means there is significance between the comparing groups at p<0.05 | ||||||||||
Also, it was shown that amongst all the plants, Citrus sinensis has the highest composition of potassium (K) in both solvents, followed by Olax subscorpioidea, Lawsonia inermis, Urena lobate and Albizia zygia in descending order. Lawsonia inermis has the highest composition of sodium (Na) in both solvents, followed by Albizia zygia, Citrus sinensis, Olax subscorpioidea and Urena lobata. Lawsonia inermis has the highest composition of calcium (Ca) in both solvents, followed by Citrus sinensis, Albizia zygia, Olax subscorpioidea and Urena lobata. For iron (Fe), Albizia zygia has the highest composition followed by Olax subscorpioidea, Citrus sinensis, Urena lobate and Lawsonia inermis. More so, Olax subscorpioidea shows the highest lead (Pb) composition followed by Urena lobata, Citrus sinensis, Albizia zygia and Lawsonia inermis. Lawsonia inermis shows the highest composition of arsenic (As) followed by Urena lobata, Albizia zygia, Citrus sinensis and Olax subscorpioidea. Likewise, Olax subscorpioidea shows the highest cadmium (Cd) composition followed by Citrus sinensis, Urena lobata, Albizia zygia and Lawsonia inermis (Table 3).
In humans, iron makes tendons and ligaments, certain chemicals of the brain are controlled by the presence or absence of iron and it is also essential for the formation of haemoglobin, which carries oxygen throughout the body70. Trace element deficits in humans can develop in many dietary situations. According to Shirin et al.71, it has been suggested that certain diseases previously thought to be incurable can potentially be treated by restoring the balance of certain elements in the human body. Recently, zinc (Zn) has shown positive effects on people suffering from atherosclerosis. There may be a relationship between Zn and its ability to fight cancer, as it is necessary for the growth and multiplication of healthy cells and the enzymes that facilitate the synthesis of nucleic acids utilize Zn72.
The study highlights the diverse phytochemical composition of medicinal plants in South-West Nigeria, indicating potential therapeutic applications. The compounds identified could be used in developing new pharmaceuticals or supplements. The findings also suggest the potential for ethnomedicine, validating traditional uses and guiding healthcare practices.
Future research should focus on the isolation and characterization of bioactive compounds and collaboration between traditional healers and modern practitioners could enhance herbal remedies' integration into mainstream medicine. Variability in plant composition and other valuable species should also be considered. Additionally, the study's focus on selected medicinal plants implies that other potentially valuable species warrant investigation.
CONCLUSION
This study highlights the diverse range of phytochemicals present in the medicinal plants studied with potential therapeutic benefits, emphasizing the significance of these plants in traditional and modern medicine. Furthermore, the elemental composition sheds light on the nutritional and elemental value of these plants, which could contribute to their holistic healing properties. This study not only expands our knowledge of the chemical constituents within medicinal plants but also lays the foundation for further studies exploring their potential applications in pharmaceuticals, nutraceuticals and holistic healthcare. Ultimately, this study underscores the importance of harnessing the inherent properties of medicinal plants to promote human well-being and drive advancements in the field of natural medicine.
SIGNIFICANCE STATEMENT
Unveiling the chemical and elemental intricacies of medicinal plants indigenous to South-West Nigeria is pivotal in unlocking the therapeutic potential embedded in traditional herbal remedies. By conducting a meticulous analysis of phytochemical constituents and elemental composition, this study not only contributes to the scientific validation of traditional medicine but also lays the foundation for the sustainable exploitation of these botanical resources. The findings hold promise for future pharmacological studies and the development of evidence-based herbal therapies, thereby fostering the integration of traditional and modern healthcare systems.
REFERENCES
- Malik, S.K., M. Ahmad and F. Khan, 2017. Qualtitative and quantitative estimation of terpenoid contents in some important plants of Punjab, Pakistan. Pak. J. Sci., 69: 150-154.
- Usin, S. and O.O. Daramola, 2022. Phytochemical analysis and evaluation of ethanol and aqueous extracts of Piliostigma thonningii leaf for in vitro antioxidant activities. Niger. J. Biochem. Mol. Biol., 37: 72-81.
- Rajput, A., R. Sharma and R. Bharti, 2022. Pharmacological activities and toxicities of alkaloids on human health. Mater. Today: Proc., 48: 1407-1415.
- Ekor, M., 2014. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol., 4.
- Favela-Hernández, J.M.J., O. González-Santiago, M.A. Ramírez-Cabrera, P.C. Esquivel-Ferriño and M.D.R. Camacho-Corona, 2016. Chemistry and pharmacology of Citrus sinensis. Molecules, 21.
- Etebu, E. and A.B. Nwauzoma, 2014. A review on sweet orange (Citrus sinensis L. Osbeck): Health, diseases and management. Am. J. Res. Commun., 2: 33-70.
- Milind, P. and C. Dev, 2012. Orange: Range of benefits. Int. Res. J. Pharm., 3: 59-63.
- Rauf, A., Ghias Uddin and J. Ali, 2014. Phytochemical analysis and radical scavenging profile of juices of Citrus sinensis, Citrus anrantifolia, and Citrus limonum. Org. Med. Chem. Lett., 4.
- Arooj, N., N. Dar and Z.Q. Samra, 2014. Stable silver nanoparticles synthesis by Citrus sinensis (orange) and assessing activity against food poisoning microbes. Biomed. Environ. Sci., 27: 815-818.
- Cardile, V., A.C.E. Graziano and A. Venditti, 2015. Clinical evaluation of Moro (Citrus sinensis(L.) Osbeck) orange juice supplementation for the weight management. Nat. Prod. Res., 29: 2256-2260.
- Liew, S.S., W.Y. Ho, S.K. Yeap and S.A.B. Sharifudin, 2018. Phytochemical composition and in vitro antioxidant activities of Citrus sinensis peel extracts. PeerJ., 6.
- Hsouna, A.B., M. Trigui, G. Culioli, Y. Blache and S. Jaoua, 2011. Antioxidant constituents from Lawsonia inermis leaves: Isolation, structure elucidation and antioxidative capacity. Food Chem., 45: 193-200.
- Majiya, H. and A. Galstyan, 2023. Potential of a methanolic extract of Lawsonia inermis (L.) leaf as an alternative sanitiser in the time of COVID-19 pandemic and beyond. J. Herb. Med., 38.
- Widyawati, T., Y.S. Pane and N.A. Yusoff, 2019. Effect of Lawsonia inermis Linn. extracts on blood glucose level in normal and streptozotocin-induced diabetic rats. Pak. J. Nutr., 18: 671-676.
- Gull, I., M. Sohail, M. Aslam and M. Athar, 2013. Phytochemical, toxicological and antimicrobial evaluation of lawsonia inermis extracts against clinical isolates of pathogenic bacteria. Ann. Clin. Microbiol. Antimicrob., 12.
- Kumar, M., P. Kaur, M. Chandel, A.P. Singh, A. Jain and S.J. Kaur, 2017. Antioxidant and hepatoprotective potential of Lawsonia inermis L. leaves against 2-acetylaminofluorene induced hepatic damage in male Wistar rats. BMC Complementary Altern. Med., 17.
- Rekik, D.M., S.B. Khedir, A. Daoud, K.K. Moalla, T. Rebai and Z. Sahnoun, 2019. Wound healing effect of Lawsonia inermis. Skin Pharmacol. Physiol., 32: 295-306.
- Lamptey, S.B. and W.K.M. Abotsi, 2017. Albizia zygia (DC.) Macbr. hydroethanol root extract exerts anti-oedemic and in vivo antioxidant activities in animal models. J. Appl. Pharm. Sci., 7: 199-205.
- Lenta, B.N., C. Vonthron-Senecheau, R.F. Soh, F. Tantangmo and S. Ngouela et al., 2007. In vitro antiprotozoal activities and cytotoxicity of some selected Cameroonian medicinal plants. J. Ethnopharmacol., 111: 8-12.
- Avoseh, O.N., F.M. Mtunzi, I.A. Ogunwande, R. Ascrizzi and F. Guido, 2021. Albizia lebbeck and Albizia zygia volatile oils exhibit anti-nociceptive and anti-inflammatory properties in pain models.J. Ethnopharmacol., 268.
- Appiah-Opong, R., I.K. Asante, D.O. Safo, I. Tuffour, E. Ofori-Attah, T. Uto and A.K. Nyarko, 2016. Cytotoxic effects of Albizia zygia (DC) J. F. MACBR, a Ghanaian medicinal plant, against human T-lymphoblast-like leukemia, prostate and breast cancer cell lines. Int. J. Pharm. Pharm. Sci., 8: 392-396.
- Odeyemi, O., A.O. Oluduro and O.M. David, 2014. Evaluation of antimicrobial activities of Albizia zygia DC leaf extracts against some clinically important pathogens. J. Nat. Sci. Res., 4: 98-105.
- Abdalla, M.A. and H. Laatsch, 2012. Flavonoids from Sudanese Albizia zygia (Leguminosae, Subfamily Mimosoideae), a plant with antimalarial potency. Afr. J. Trad. Complementary Altern. Med., 9: 56-58.
- Okpo, S.O., S.M. Kporha and M.C. Chijioke, 2016. Gastro-protective potential of aqueous stem bark extract of Albyzia zygia in rat models of ulcer. J. Pharm. Allied Sci., 13: 2309-2318.
- Abotsi, W.K.M., S.B. Lamptey, E. Boakye-Gyasi and E. Woode, 2017. Albizia zygia (DC.) J.F. Macbr. (Leguminosae-Mimosoideae) root extract exhibits anti-nociceptive and antipyretic activities in murine models. J. Ethnopharmacol., 199: 183-193.
- Amoateng, P., D. Osei-Safo, K.K.E. Kukuia, S. Adjei, C. Agbemelo-Tsomafo, S.N. Adu-Poku and K.Y. Agyeman-Badu, 2017. Psychotropic effects of an alcoholic extract from the leaves of Albizia zygia (Leguminosae-Mimosoideae). Evidence-Based Complementary Altern. Med., 2017.
- Kumbol, V.W.A., W.K.M. Abotsi and R.P. Biney, 2021. Antidepressant-like effect of Albizia zygia root extract in murine models. J. Basic Clin. Physiol. Pharmacol., 32.
- Oloyede, G.K. and A.O. Ogunlade, 2013. Phytochemical screening, antioxidant, antimicrobial and toxicity activities of polar and non-polar extracts of Albizia zygia (DC) stem-bark. Annu. Res. Rev. Biol., 3: 1020-1031.
- Okpo, S.O., C.O. Igwealor and G.I. Eze, 2016. Sub-acute toxicity study on the aqueous extract of Albizia zygia stem bark. J. Pharm. Bioresour., 13: 32-41.
- Babu, S.S., D.B. Madhuri and S.L. Ali, 2016. A pharmacological review of Urena lobata plant. Asian J. Pharm. Clin. Res., 9: 20-22.
- Sajem, A.L. and K. Gosal, 2006. Traditional use of medicinal plants by the Jaintia tribes in North Cachar Hills district of Assam, Northeast India. J. Ethnobiol. Ethnomed., 2.
- Pieme, C.A., V.N. Penlap, J. Ngogang and M. Costache, 2010. In vitro cytotoxicity and antioxidant activities of five medicinal plants of Malvaceae family from Cameroon. Environ. Toxicol. Pharmacol., 29: 223-228.
- Gao, X.L., Y. Liao, J. Wang, X.Y. Liu and K. Zhong et al., 2015. Discovery of a potent anti-yeast triterpenoid saponin, clematoside-S from Urena lobata L. Int. J. Mol. Sci., 16: 4731-4743.
- Islam, M.T., T.A. Riaz, S.A. Ayatollahi and J. Sharifi-Rad, 2021. Anxiolytic-like effect of Urena lobata (L.) in swiss albino mice. Clin. Phytosci., 7.
- Fagbohun, E.D., R.R. Asare and A.O. Egbebi, 2012. Chemical composition and antimicrobial activities of Urena lobata L. (Malvaceae). J. Med. Plants Res., 6: 2256-2260.
- Xiao-Ping, S., S. Cong, Q. Bo-Wen, Y. Wan-Qing and W. Yun et al., 2017. Flavonoid glycosides from aerial part of Urena lobata. Chin. Pharm. J., 52: 1670-1674.
- Su, C., B. Qi, J. Wang, N. Ding and Y. Wu et al., 2018. Megastigmane glycosides from Urena lobata. Fitoterapia, 127: 123-128.
- Luo, Y., C. Su, N. Ding, B. Qi and F. Jia et al., 2019. Lignan glycosides from Urena lobata. Molecules, 24.
- Odoma, S., A.U. Zezi, N.M. Danjuma, A. Ahmed and M.G. Magaji, 2017. Elucidation of the possible mechanism of analgesic actions of butanol leaf fraction of Olax subscorpioidea Oliv. J. Ethnopharmacol., 199: 323-327.
- Adeoluwa, O.A., A.O. Aderibigbe and E.T. Olonode, 2014. Antinociceptive property of Olax subscorpioidea Oliv (Olacaceae) extract in mice. J. Ethnopharmacol., 156: 353-357.
- Orabueze, I.C., A.A. Amudalat and A.A. Usman, 2016. Antimicrobial value of Olax subscorpioidea and Bridelia ferruginea on micro-organism isolates of dental infection. J. Pharmacogn. Phytochem., 5: 398-406.
- Gbadamosi, I.T., L.A. Raji, A.A. Oyagbemi and T.O. Omobowale, 2017. Hypolipidemic effects of Olax subscorpioidea Oliv. root extract in experimental rat model. Afr. J. Biomed. Res., 20: 293-299.
- Ibukunoluwa, M.R., 2017. In vivo anti-plasmodial activity and histopathological analysis of water and ethanol extracts of a polyherbal antimalarial recipe. J. Pharmacogn. Phytother., 9: 87-100.
- Okoli, R.I., O. Aigbe, J.O. Ohaju-Obodo and J.K. Mensah, 2007. Medicinal herbs used for managing some common ailments among Esan people of Edo State, Nigeria. Pak. J. Nutr., 6: 490-496.
- Ibukunoluwa, M.R., T.A. Olusi and E.O. Dada, 2015. Assessment of chemical compositions of three antimalarial plants from Akure, Southwestern Nigeria: A preliminary study. Afr. J. Plant Sci., 9: 313-319.
- Wisdom, N.N., E.E. Bassey, F.B. Jelani, G.A. Ishaku, U.M. Uwem and S.C. Joseph, 2016. Biochemical studies of Ocimum sanctum and Olax subscorpioidea leaf extracts. J. Pharm. Res. Int., 12.
- Molyneux, R.J., S.T. Lee, D.R. Gardner, K.E. Panter and L.F. James, 2007. Phytochemicals: The good, the bad and the ugly? Phytochemistry, 68: 2973-2985.
- Afendi, F.M., T. Okada, M. Yamazaki, A. Hirai-Morita and Y. Nakamura et al., 2012. KNApSAcK family databases: Integrated metabolite-plant species databases for multifaceted plant research. Plant Cell Physiol., 53.
- Rutz, A., M. Sorokina, J. Galgonek, D. Mietchen and E. Willighagen et al., 2022. The LOTUS initiative for open knowledge management in natural products research. eLife, 11.
- Shibamoto, T. and L.F. Bjeldanes, 2009. Introduction to Food Toxicology. 2nd Edn., Academic Press, Cambridge, Massachusetts, ISBN: 9780080921532, Pages: 320.
- Shaw, D., 2010. Toxicological risks of Chinese herbs. Planta Med., 76: 2012-2018.
- Halliwell, B., 2007. Dietary polyphenols: good, bad, or indifferent for your health? Cardiovasc. Res., 73: 341-347.
- Balogun, I.O. and O.P. Olatidoye, 2012. Chemical composition and nutritional evaluation of velvet bean seeds (Mucuna utilis) for domestic consumption and industrial utilization in Nigeria. Pak. J. Nutr., 11: 116-122.
- Paul, B.K., M.M. Saleh-E-In, A. Ara and S.K. Roy, 2013. Minerals and nutritional composition of Radhuni (Carum roxburghianum Benth.) seeds. Int. Food Res. J., 20: 1731-1737.
- Daramola, O.O., S.G. Usin, U.E. Okon and O.M. Olugbenga, 2022. Antibacterial properties and phytochemicals screening of Tectona grandis (Teak) leaf extracts against bacteria implicated from upper respiratory tract infections. J. Microbes Res., 1.
- Sani, Y.M., A.M. Musa, U.U. Pateh, A.K. Haruna and A.H. Yaro et al., 2014. Phytochemical screening and preliminary evaluation of analgesic and anti-inflammatory activities of the methanol root extract of Cissus polyantha. Bayero J. Pure Appl. Sci., 7: 19-23.
- Harborne, J.B., 1984. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis. 2nd Edn., Chapman and Hall, London, United Kingdom., ISBN-13: 9780412255502, Pages: 288.
- Obadoni, B.O. and P.O. Ochuko, 2002. Phytochemical studies and comparative efficacy of the crude extracts of some haemostatic plants in Edo and Delta States of Nigeria. Global J. Pure Appl. Sci., 8: 203-208.
- Ejikeme, C.M., C.S. Ezeonu and A.N. Eboatu, 2014. Determination of physical and phytochemical constituents of some tropical timbers indigenous to Niger Delta area of Nigeria. Eur. Sci. J., 10: 247-270.
- Kaur, G.J. and D.S. Arora, 2009. Antibacterial and phytochemical screening of Anethum graveolens, Foeniculum vulgare and Trachyspermum ammi. BMC Complementary Altern. Med., 9.
- Ezeonu, C.S. and C.M. Ejikeme, 2016. Qualitative and quantitative determination of phytochemical contents of indigenous Nigerian softwoods. New J. Sci., 2016.
- Bussmann, R.W., L. Hennig, A. Giannis, J. Ortwein, T.M. Kutchan and X. Feng, 2013. Anthraquinone content in noni (Morinda citrifolia L.). Evidence-Based Complementary Altern. Med., 2013.
- Adelantado, J.V.G., V.P. Martinez, A.P. Garcia and F.B. Reig, 1991. Atomic-absorption spectrometric determination of calcium, magnesium and potassium in leaf samples after decomposition with molten sodium hydroxide. Talanta, 38: 959-963.
- Chan, K.W., S. Iqbal, N.M.H. Khong, D.J. Ooi and M. Ismail, 2014. Antioxidant activity of phenolics-saponins rich fraction prepared from defatted kenaf seed meal. LWT-Food Sci. Technol., 56: 181-186.
- Akbari, S., N.H. Abdurahman and R.M. Yunus, 2019. Optimization of saponins, phenolics, and antioxidants extracted from fenugreek seeds using microwave-assisted extraction and response surface methodology as an optimizing tool. C. R. Chim., 22: 714-727.
- Panche, A.N., A.D. Diwan and S.R. Chandra, 2016. Flavonoids: An overview. J. Nutr. Sci., 5.
- Martins, N., L. Barros, C. Santos-Buelga, M. Henriques, S. Silva and I.C.F.R. Ferreira, 2015. Evaluation of bioactive properties and phenolic compounds in different extracts prepared from Salvia officinalis L. Food Chem., 170: 378-385.
- de Andrade Monteiro, C. and J.R.A. dos Santos, 2019. Phytochemicals and Their Antifungal Potential Against Pathogenic Yeasts. In: Phytochemicals in Human Health, Rao, A.V., D.R.A. Mans and L. Rao (Eds.), IntechOpen, London, United Kingdom, ISBN: 978-1-78985-588-3 pp: 1-31.
- Usin, S.G., O.O. Daramola, O.M. Olugbenga, F. Ayobami, O.D. Rotimi, A.B. Christainah and D.A. Elizabeth, 2023. Phytochemical analysis, mineral composition and antimicrobial activities of ethyl acetate and ethanol extracts of Vernonia amygdalina and Morinda lucida against some clinical isolated pathogenic microorganisms: A comparative study. J. Med. Herbs, 13: 55-66.
- Akodu, O.S., E.A. Disu, O.F. Njokanma and O.A. Kehinde, 2016. Iron deficiency anaemia among apparently healthy pre-school children in Lagos, Nigeria. Afr. Health Sci., 16: 61-68.
- Shirin, K., S. Imad, S. Shafiq and K. Fatima, 2010. Determination of major and trace elements in the indigenous medicinal plant Withania somnifera and their possible correlation with therapeutic activity. J. Saudi Chem. Soc., 14: 97-100.
- Caballero-George, C., P.M.L. Vanderheyden, P.N. Solis, L. Pieters and A.A. Shahat et al., 2001. Biological screening of selected medicinal panamanian plants by radioligand-binding techniques. Phytomedicine, 8: 59-70.
How to Cite this paper?
APA-7 Style
Usin,
S.G., Okon,
U.E., Daramola,
A.E., Emmanuel,
P.D. (2024). Evaluation of Phytochemical Constituents and Elemental Profiling of Selected Medicinal Plants in South-West, Nigeria. Asian Journal of Biological Sciences, 17(1), 145-155. https://doi.org/10.3923/ajbs.2024.145.155
ACS Style
Usin,
S.G.; Okon,
U.E.; Daramola,
A.E.; Emmanuel,
P.D. Evaluation of Phytochemical Constituents and Elemental Profiling of Selected Medicinal Plants in South-West, Nigeria. Asian J. Biol. Sci 2024, 17, 145-155. https://doi.org/10.3923/ajbs.2024.145.155
AMA Style
Usin
SG, Okon
UE, Daramola
AE, Emmanuel
PD. Evaluation of Phytochemical Constituents and Elemental Profiling of Selected Medicinal Plants in South-West, Nigeria. Asian Journal of Biological Sciences. 2024; 17(1): 145-155. https://doi.org/10.3923/ajbs.2024.145.155
Chicago/Turabian Style
Usin, Saviour, Godswealth, Unwana Ema Okon, Adedoyin Elizabeth Daramola, and Praise Dyap Emmanuel.
2024. "Evaluation of Phytochemical Constituents and Elemental Profiling of Selected Medicinal Plants in South-West, Nigeria" Asian Journal of Biological Sciences 17, no. 1: 145-155. https://doi.org/10.3923/ajbs.2024.145.155

This work is licensed under a Creative Commons Attribution 4.0 International License.



