Detection of Plasmodium falciparum Kelch 13 Propeller Gene Polymorphism Among Outpatients in Katsina State, Nigeria
| Received 18 Jan, 2024 |
Accepted 17 Apr, 2024 |
Published 30 Sep, 2024 |
Background and Objective: Malaria case management relies on Artemisinin Combination Therapy (ACT), but the emergence of artemisinin-resistant Plasmodium falciparum poses a threat to global malaria elimination. Kelch 13 gene mutations are associated with artemisinin resistance and their monitoring is crucial for a timely response. Nigeria, with the highest malaria burden in Africa, faces the risk of artemisinin-resistant parasites due to travel patterns. The study focuses on detecting K13 gene polymorphism in Katsina Nigeria to prevent drug resistance and inform treatment policies. Materials and Methods: The study, conducted in three Local Governments of Katsina State, collected 210 samples from three general hospitals. The DNA was extracted from P. falciparum-positive samples and the K13 gene was amplified using nested Polymerase Chain Reaction (PCR). Sequencing and phylogenetic analysis were performed to identify gene variants and assess evolutionary relationships. Statistical analysis evaluated prevalence variations among hospitals. Results: Prevalence analysis revealed significant variations among three Local Government Areas, with Dutsin-ma having the highest (42.86%) and Ingawa the lowest (18.57%). The fragment of propeller that was investigated shows the presence of polymorphism among outpatients in three Local Government Areas of Katsina State. Four polymorphic sites were discovered in the propeller region of this gene. However, none of the four most implicated mutations associated with ACT resistance (Y493H, R539T, I543T or C580Y) were seen in the study area. Conclusion: The phylogenetic analysis revealed a common origin for all four identified genes within the same domain, indicating a shared ancestry.
| Copyright © 2024 Amiru et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Malaria remains the most deadly parasitic diseases especially in the tropics and subtropics, causing 229 million cases and approximately 409,000 death globally in 20191. Regardless of the efforts to curtail the incident of malaria and death through diagnosis and treatment, malaria still poses a serious risk to public health. The proliferation of parasites resistant to drugs has been the primary factor contributing to the effect of malaria seen in recent years especially in the tropics2.
Resistant to artemisinin is related to several recorded polymorphisms of the Kelch 13 gene, these resistance genes have been reported in some African parasite populations3. Nigeria has the leading malaria burden in Africa, accounting for approximately 27% of the worldwide malaria burden4. There is an emerging fear that artemisinin-resistant strains could emerge in Africa, as seen in some years past in the case of chloroquine and sulphadoxine/pyrimethamine-resistant parasites as a result of the global network and connectivity to other countries, especially the Asian nations. Hence, monitoring the genes that cause resistance is important to detect and address the issue of resistance quickly. Furthermore, any probable impact of artemisinin resistance on the current malaria control program on artemisinin-based combination therapies may have deadly consequences on the total malaria elimination target.
In Nigeria, ACT was first used in 2005, with artesunate plus amodiaquine serving as the backup treatment for uncomplicated malaria and Artemether-Lumefantrine (AL) serving as the first-line treatment. Artemisinin derivatives, which include artesunate, artemether and dihydroartemisinin (DHA), are well-tolerated, fast-acting medications that are frequently used in conjunction with longer-acting partner medicines, or ACTs. Because ART derivatives have a short half-life (usually less than 1 hr) in plasma, longer-acting partner medications that can eradicate leftover parasites after the ART component has fallen to subtherapeutic concentrations must be used5.
The Kelch family of proteins which Kelch 13 belongs to has several protein-to-protein contact locations and also mediates several cellular functions as well as protein degradation and oxidative stress reactions6. These genes are close to areas related to slow parasite clearance rates and are located on the chromosome 13 gene of the P. falciparum7,8. These genomes are found in all developmental stages of P. falciparum, the trophozoites, the schizonts (asexual stage) as well as the gametocytes (sexual stage). The alterations at a particular gene code on the K13 are associated with sluggish clearance of infection and hence used as an indicator for resistance in artemisinin. Modification in the transporter gene, the protein sequence alters the physiochemical qualities and functional character of the genomes. Each protein sequence has unique and precise qualities that affect its mode of action, any alterations are associated with drug resistance in malaria9. This study determine the prevalence of Plasmodium falciparum parasite among the three Local Government Areas, where the genes responsible for artemisinin resistance were identify and amplify to determine the different mutations between the genes.
MATERIALS AND METHODS
Study area: This study was conducted in three Local Governments Areas of Katsina State between August and September of 2022. These include Dutsin-Ma Local Government Area, which is located between Latitudes 12°17.00N to 12°17.84 N and Longitude 7°0'26 E and has a land size of approximately 552.323 km2 with a population of approximately 169,826 as at the 2006 national population census, Kankia Local Government Area, which is a town with an area of 824 km2 and a human population of 151,434 and Ingawa Local Government Area which has its geographical coordinates 12°38'35"N to 8°2'59"E with an area of 892 km2 and population of 169,75310.
Sample collection: A total of 210 samples were collected from outpatients attending three general hospitals in the study area. Seventy samples were collected in triplicate from each hospital, representing individuals who exhibited clinical symptoms of malaria. Venous blood was collected from patients attending the hospital for diagnosis. Blood samples were immediately screened using HRP2-based RDT kits (Care Start®, Access Bio Inc., Batch number NJ, USA) and samples found positive were used for preparation of thick and thin smears and later taken to the laboratory for diagnosis by microscopy. A few drops of blood positive for malaria were spotted on Whatman® no. 3 mm filter paper (GE Healthcare Life Sciences) (three spots per patient) and allowed to dry at room temperature. Each filter paper was kept in a separate zip-locked sachet with silica gel, stored at room temperature as described by Oboh et al.11 and used for molecular diagnosis using the Kelch13 gene's Deoxyribonucleic Acid (DNA) sequencing and nested Polymerase Chain Reaction (PCR).
DNA extraction: Sixty five Dried Blood Spots (DBS) confirmed to be positive for P. falciparum were used in this investigation. Using a single-hole paper puncher, 3 mm diameter punches were formed from the dried blood spot. These were put into a 1.5 mL microcentrifuge tube and using the QIAamp_DNA Mini kit, the genomic DNA from each DBS was extracted in accordance with the manufacturer's instructions (Qiagen, Germany).
Polymerase chain reaction and sequencing: Nested PCR was done using oligonucleotide primers K13 (P) F: 5´-GGGAATCTGGTGGTAACAGC-3´ R: 5´-CGGAGTGACCAAATCTGGGA-3´and cycling conditions of 95°C 15 min; {95°C 30 sec, 58°C 2 min, 72°C 2 min} 30 cycles; 72°C 10 min, Nested PCR: K13 (S) F: 5´-GCCTTGTTGAAAGAAGCAGA-3´, R: 5´GCCAAGCTGCCATTCATTTG-3´ and cycling conditions of 95°C 15 min; (95°C 30 min, 60°C 1 min, 72°C 1 min) 40 cycles; 72°C 10 min, respectively.
A total of 2 μL of template genomic DNA generated from the dried blood spots, 1 μL each of primer F and R, 12.5 μL of 2*Taq DNA master mix and 8.5 μL of double-distilled water (ddH2O) made up the 25 μL amplification reaction mixture were used. The results of the nested PCR were sequenced using an ABI 3130xl Genetic analyzer (Applied Biosystems, Foster City, California, USA) after being seen on 1% agarose gel using a Bio-Rad molecular imager(R) Gel DocTM. Using the reference P. falciparum K13 gene sequence (PF3D7_134700), BLAST search (www.ncbi.nlm.nih.gov) was used to perform sequence alignment and analysis.
RESULTS
Prevalence of Plasmodium falciparum among three local government areas: The prevalence of Plasmodium falciparum parasites from the three local government areas were presented in Table 1. In Dutsin-Ma Local Government Area, having the highest prevalence of Plasmodium falciparum recorded the mean positive cases were 30, while the mean negative cases were 40, resulting in a percentage prevalence of 42.86%. This is followed by Kankia Local Government Area, with the mean positive and negative cases of 22 and 48, respectively, making a percentage prevalence of 31.43%. The lowest prevalence of Plasmodium falciparum was recorded from Ingawa Local Government Area, reporting 13 mean positive cases, 57 mean negative cases and a percentage prevalence of 18.57%. The total across all Dutsin-Ma, Kankia and Ingawa Local Government Areas showed 65 mean positive cases, 145 mean negative cases and an overall percentage prevalence of 92.86% as presented in Table 1.
The Analysis of Variance (ANOVA) conducted to assess the prevalence of Plasmodium falciparum parasites among three local government areas shows significant differences. The associated p-value was highly significant (p = 0.0016), indicating that there is a statistically significant difference in parasite prevalence among the three Local Government as presented in Table 2. Figure 1 was the Agrose gel of PCR for amplification of Kelch 13 fragment and Fig. 2 was the polygenetic tree.
| Table 1: | Prevalence of Plasmodium falciparum among three local government areas | |||
| Hospital | Mean positive | Mean negative | Prevalence (%) |
| GHD | 30 | 40 | 42.86 |
| GHK | 22 | 48 | 31.43 |
| GHI | 13 | 57 | 18.57 |
| Total | 65 | 145 | 92.86 |
| GHD: General Hospital Dutsin-Ma, GHK: General Hospital Kankia and GHI: General Hospital Ingawa | |||
| Table 2: | Analysis of variance on the prevalence of Plasmodium falciparum parasites among three hospitals | |||
| Source of variation | SS | Df | MS | F | p-value | F-critical |
| Between hospitals | 434 | 2 | 217 | 22.44828 | 0.001638 | 5.143253 |
| Within hospitals | 58 | 6 | 9.666667 | |||
| Total | 492 | 8 | ||||
| SS: Sum square, Df: Degree of freedom, MS: Mean square and F: Calculated F-value | ||||||
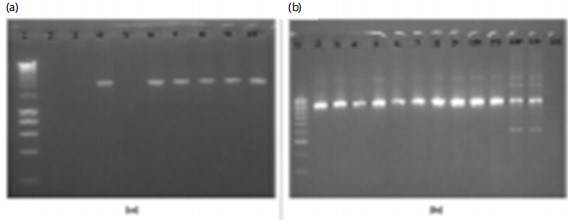
|

|
DISCUSSION
The data presented in Table 1 clearly demonstrate significant variations in the prevalence of Plasmodium falciparum across the three Local Government Areas. Dutsin-Ma L.G.A exhibited the highest prevalence, with a percentage of 42.86%, followed by Kankia at 31.43% and Ingawa with the lowest prevalence at 18.57%. These variations could be attributed to several factors, including environmental conditions, socio-economic factors and healthcare infrastructure.
The geographic location of each hospital may play a significant role in malaria prevalence. Factors such as temperature, humidity and proximity to water bodies can influence the breeding and survival of the Anopheles mosquitoes, which are the vectors for Plasmodium falciparum. Academic research has shown that environmental conditions have a direct impact on the prevalence and transmission of malaria12.
Another reason could be that socio-economic conditions, including housing quality, access to healthcare and educational levels, can also contribute to malaria prevalence. Communities with lower socio-economic status may face challenges in implementing preventive measures such as bed net usage and access to antimalarial medications. Studies by Lengeler13 have demonstrated the association between socio-economic factors and malaria incidence. Disparities in healthcare infrastructure and access to diagnostic and treatment facilities may contribute to variations in reported malaria cases.
The analysis identified four distinct Kelch 13 propeller genes, namely K13_Target 1, K13_Target 2, K13_Target 3 and K13_Target 4, each with different base pair lengths. K13_Target 1 comprises 814bp, K13_Target 2 has 861bp, K13_Target 3 has 820bp and K13_Target 4 has 805bp. It is noteworthy that these lengths are significantly shorter than the reference K13 gene, which has a length of 2181bp. The variations in gene lengths suggest potential genetic mutations or alterations that may be associated with artemisinin resistance.
The phylogenetic analysis revealed a common origin for all four identified genes within the same domain, indicating a shared ancestry. However, notable differences in evolutionary relationships were observed. Target 2 and the reference K13 gene appeared to share a closer evolutionary relationship, forming a distinct branch in the phylogenetic tree. This observation is of particular significance as it may indicate a potential link between the artemisinin resistance-associated mutations in Target 2 and those in the reference K13 gene.
Furthermore, the connectivity observed in the final branch of the phylogenetic tree, with both Target 2 and the reference K13 gene connected to Target 1, suggests a shared genetic history and potential transfer of genetic material between these variants. On the other hand, Target 3 exhibited the least genetic relation, indicating a divergence in its evolutionary path from the other three targets.
The analysis of the K13 gene across the four K13 Target samples reveals intriguing insights into the conservation patterns within this crucial gene associated with artemisinin resistance in Plasmodium falciparum. The high level of conservation, with over 95% identity at each position, highlights the overall stability of the K13 gene in the studied samples. This finding aligns with previous studies emphasizing the importance of the K13 gene in mediating resistance to artemisinin-based therapies14.
While the analysis identifies minor variations such as single nucleotide polymorphisms (SNPs) and one insertion/deletion (indel) polymorphism, these changes are deemed relatively inconsequential. This observation is consistent with the notion that the K13 gene is under selective pressure to maintain its functional integrity, limiting the tolerance for significant alterations that might compromise its role in artemisinin resistance8.
The most noteworthy aspect of the findings is the pronounced conservation observed in the first and third exons of the K13 gene. These exons encode the functional domains of the K13 protein, which are crucial for conferring resistance to artemisinin against Plasmodium falciparum. The robust conservation in these exons implies a strong selective pressure acting on them, underscoring their indispensability for the proper functioning of the K13 protein. This aligns with the understanding that mutations in these domains are associated with the development of artemisinin resistance15.
Conversely, the second exon of the K13 gene displays lower conservation, marked by a slightly higher occurrence of SNPs and indels. This suggests reduced selective pressure on this exon, potentially indicating a greater tolerance for genetic variation. However, it is crucial to note that even within this exon, an overall high level of conservation persists, exceeding 90% identity. This may imply that while the second exon can tolerate some genetic variation, there are still constraints imposed by functional requirements.
This study recommended that the Intensify malaria surveillance efforts in Katsina State, with a focus on monitoring and understanding the prevalence of Plasmodium falciparum. The Implement routine screening and molecular surveillance to detect Kelch 13 polymorphisms, providing valuable data for timely intervention. An increase accessibility to diagnostic tools, such as molecular techniques like DNA sequencing, for more accurate detection of resistance markers are required.
Limitation of this study was the small sample size and restricted study areas are insufficient to identify newly emergent resistance loci in the populations, which may be present at low frequencies. The restricted availability of collected DNA and RNA prevented us from verifying the identification of the putative mutant kelch 13 gene or verify its existence via sequencing techniques.
CONCLUSION
This research detect four Plasmodium falciparum Kelch 13 propeller gene polymorphism in patients attending three hospitals in Katsina State, Nigeria. The study revealed significant variations in the prevalence of Plasmodium falciparum across three Local Government Areas in the state, with Dutsin-Ma exhibiting the highest prevalence. The genetic analysis identified four distinct Kelch 13 propeller genes, suggesting potential genetic mutations associated with artemisinin resistance but none among those validated genes associated with ACT resistance (Y493H, R539T, I543T or C580Y).
SIGNIFICANCE STATEMENT
The prevalence of drug-resistant Plasmodium falciparum has increased malaria-related mortality, particularly in Sub-Saharan Africa. By learning more about the molecular markers that are responsible for this phenomena makes it more efficient in monitoring the increase of drug-resistant parasites in a given region. To do this, it's critical to apply molecular methods like DNA sequencing, which can identify the many gene changes causing resistance to the antimalarial drugs that are now available. The genetic analysis identified four distinct Kelch 13 propeller genes, suggesting potential genetic mutations associated with artemisinin resistance.
REFERENCES
- Tuteja, R., 2007. Malaria-An overview. FEBS J., 274: 4670-4679.
- Tola, M., O. Ajibola, E.T. Idowu, O. Omidiji, S.T. Awolola and A. Amambua-Ngwa, 2020. Molecular detection of drug resistant polymorphisms in Plasmodium falciparum isolates from Southwest, Nigeria. BMC Res. Notes, 13.
- Igbasi, U., W. Oyibo, S. Omilabu, H. Quan and S.B. Chen et al., 2019. Kelch 13 propeller gene polymorphism among Plasmodium falciparum isolates in Lagos, Nigeria: Molecular epidemiologic study. Trop. Med. Int. Health, 24: 1011-1017.
- Aribodor, D.N., I.K. Ugwuanyi and O.B. Aribodor, 2016. Challenges to achieving malaria elimination in Nigeria. Am. J. Public Health Res., 4: 38-41.
- Eastman, R.T. and D.A. Fidock, 2009. Artemisinin-based combination therapies: A vital tool in efforts to eliminate malaria. Nat. Rev. Microbiol., 7: 864-874.
- Adams, J., R. Kelso and L. Cooley, 2000. The kelch repeat superfamily of proteins: Propellers of cell function. Trends Cell Biol., 10: 17-24.
- Cheeseman, I.H., B.A. Miller, S. Nair, S. Nkhoma and A. Tan et al., 2012. A major genome region underlying artemisinin resistance in malaria. Science, 336: 79-82.
- Mohon, A.N., M. Shafiul Alam, A.G. Bayih, A. Folefoc, D. Shahinas, Rashidul Haque and D.R. Pillai, 2014. Mutations in Plasmodium falciparum K13 propeller gene from Bangladesh (2009-2013). Malar. J., 13.
- Ariey, F., B. Witkowski, C. Amaratunga, J. Beghain and A.C. Langlois et al., 2014. A molecular marker of artemisinin-resistant Plasmodium falciparum malaria. Nature, 505: 50-55.
- Omojuyigbe, J.O., A.J.J. Owolade, T.O. Sokunbi, H.A. Bakenne, B.A. Ogungbe, H.J. Oladipo and P.I. Agughalam, 2023. Malaria eradication in Nigeria: State of the nation and priorities for action. J. Med. Surg. Public Health, 1.
- Oboh, M.A., A.S. Badiane, G. Ntadom, Y.D. Ndiaye, K. Diongue, M.A. Diallo and D. Ndiaye, 2018. Molecular identification of Plasmodium species responsible for malaria reveals Plasmodium vivax isolates in Duffy negative individuals from Southwestern Nigeria. Malar. J., 17.
- Hay, S.I., D.J. Rogers, G.D. Shanks, M.F. Myers and R.W. Snow, 2001. Malaria early warning in Kenya. Trends Parasitol., 17: 95-99.
- Lengeler, C., 2004. Insecticide-treated bed nets and curtains for preventing malaria. Cochrane Database Syst. Rev., 2004.
- Ménard, D., N. Khim, J. Beghain, A.A. Adegnika and M. Shafiul-Alam et al., 2016. A worldwide map of Plasmodium falciparum K13-propeller polymorphisms. N. Engl. J. Med., 374: 2453-2464,
- Abubakar, U.F., R. Adam, M.M. Mukhtar, A. Muhammad, A.A. Yahuza and S.S. Ibrahim, 2020. Identification of mutations in antimalarial resistance gene Kelch13 from Plasmodium falciparum isolates in Kano, Nigeria. TropicalMed, 5.
How to Cite this paper?
APA-7 Style
Amiru,
A., Eberemu,
N.C., Benshima,
O.J. (2024). Detection of Plasmodium falciparum Kelch 13 Propeller Gene Polymorphism Among Outpatients in Katsina State, Nigeria. Asian Journal of Biological Sciences, 17(3), 307-313. https://doi.org/10.3923/ajbs.2024.307.313
ACS Style
Amiru,
A.; Eberemu,
N.C.; Benshima,
O.J. Detection of Plasmodium falciparum Kelch 13 Propeller Gene Polymorphism Among Outpatients in Katsina State, Nigeria. Asian J. Biol. Sci 2024, 17, 307-313. https://doi.org/10.3923/ajbs.2024.307.313
AMA Style
Amiru
A, Eberemu
NC, Benshima
OJ. Detection of Plasmodium falciparum Kelch 13 Propeller Gene Polymorphism Among Outpatients in Katsina State, Nigeria. Asian Journal of Biological Sciences. 2024; 17(3): 307-313. https://doi.org/10.3923/ajbs.2024.307.313
Chicago/Turabian Style
Amiru, Abdulrahman, Nkiru Charity Eberemu, and Orpin James Benshima.
2024. "Detection of Plasmodium falciparum Kelch 13 Propeller Gene Polymorphism Among Outpatients in Katsina State, Nigeria" Asian Journal of Biological Sciences 17, no. 3: 307-313. https://doi.org/10.3923/ajbs.2024.307.313

This work is licensed under a Creative Commons Attribution 4.0 International License.



